Research Article
Antimicrobial Activity of the Methanolic and Crude Alkaloid Extracts of Acalypha wilkesiana cv. macafeeana Copper Leaf
Department of Biosciences and Biotechnology,Babcock University, PMB 21244, Ikeja, Lagos 100 001, Nigeria
C.P. Anokwuru
Department of Chemical and Environmental Sciences, Babcock University, PMB 21244, Ikeja, Lagos 100 001, Nigeria
E. Nsofor
Department of Chemical and Environmental Sciences, Babcock University, PMB 21244, Ikeja, Lagos 100 001, Nigeria
O.A. Odusanya
Department of Chemical and Environmental Sciences, Babcock University, PMB 21244, Ikeja, Lagos 100 001, Nigeria
O. Adebanjo
Department of Biosciences and Biotechnology,Babcock University, PMB 21244, Ikeja, Lagos 100 001, Nigeria









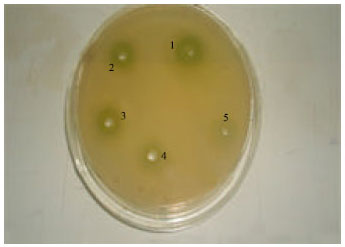


larry l ball Reply
If I understand you were trying to determine if this leaf extract would inhibit the mold growth to inhiubit the spread of staff and other molds harmful to humans. In essence your determination was that the extract had minimal affect on the different molds subjected to the extract.keep trying some type of leaf extract may do a better job.