ABSTRACT
The present study is an attempt to clarify the role of captive dogs as a source of some zoonotic bacteria to their contacts or vise versa. Bacteriological examination of fecal swabs evidenced infection by 3 enteric bacteria in attendants, puppies and dogs. Salmonella (20, 33.3 and 41.67%), Campylobacter (13.33, 33.3 and 33.3%) and Enteroinvasive E. coli (46.66, 46.67 and 58.33%). Serotyping of these bacteria revealed presence of S. typhimurium in dogs (60%) and attendants (66.67%), S. enteritidis in one of the worker as well as four untyped strains. Two serotypes of Campylobacter as C. jejuni in two workers and four dogs, C. coli in three dogs, while two untyped isolates were recorded in dogs. Three serotypes of E. coli (O 26, O 76 and O 55) and two untyped strains were isolated from workers and dogs. Moreover two isolates (O 5 and O 111) were diagnosed from dogs only. The isolates showed high sensitivity for Gentamycin (10 μg) and Tetracyclin (30 μg). The study recommended some precautionary measures to minimize the role of captive dogs as a potential source of zoonotic pathogens.
PDF Abstract XML References Citation
How to cite this article
URL: https://scialert.net/abstract/?doi=jm.2009.222.228
INTRODUCTION
Through out their long history of domestication, dogs have been source of zoonotic pathogens and have served as a link for diseases exchange among livestock, wildlife and humans. A number of potential enteropathogens have been isolated from dogs, most notably Salmonella sp. and Campylobacter sp., as well as Enteroinvasive E. coli (EIEC). These pathogens represent an interesting combination of incidents, severity, stringency of industry standards and public awareness (Greene, 2006). Meanwhile, they have been clearly associated with acute and chronic disease, typically causing enterocolitis, but they can also be present in clinically healthy carriers which potentially present a risk to other animals and humans so that they represent a zoonotic risk (Hall and Simpson, 2000).
Bacterial diarrheas are generally a nuisance for the adult animal but can be lethal to a small puppy, kitten, or even a human baby. Most of these problems stem from contaminated food or fecal contaminated environment. Raw food diets for pets dramatically increase the risk of human exposure (Brook, 2006). Dogs act as carriers for many enteropathogens and the development of clinical disease on the infected animal may involve an environmental factors such as stress and an innate inability to mount an effective mucosal immune response. Moreover, dogs can play a role of active carriers that tend to shed organisms continuously or intermittently or as latent carriers shed pathogens only when under stress (Batt, 2002).
The survival time of these bacteria varies significantly, Campylobacter, for example can survive for days in surface water and as long as four weeks in feces. In addition, the duration of excretion in infected dogs and cats can be as long as four months. Salmonellae can also survive for prolonged periods outside the host, especially in aquatic environments; shedding may continue for 3 to 6 weeks and can be reactivated at a later date by any intercurrent stress (Hall, 2004).
In Egypt, no recent data about the incidence of different bacteria infecting dogs are available. However, several studies concerning enteric bacteria infecting human, food and water have been published. Wierzba et al. (2006) reported a percentage of 46% of pediatric diarrhea in Egyptian 2-6 years old children in the Nile River Delta. They were infected by one or more pathogens including, rotavirus, enterotoxigenic Escherichia coli (ETEC) and Campylobacter. Bacteria-associated diarrhea was prevalent during summer. Children with Shigella- or Campylobacter-associated diarrhea reported as watery diarrhea and rarely dysentery. The ETEC did not have any clinically distinct characteristics. Their study suggested that rotavirus is of principle concern, followed by ETEC, Shigella and Campylobacter. Fareed et al. (2008) isolated eight bacterial genera from Manzalah Lake water at North-East of Egypt. These organisms are Escherichia coli (20%), Pseudomonas aeruginosa (16%), Klebsiella pneumoniae (14%), Salmonella colerasuis (13%), Serratia liquefaciens (10%), Shigella sp., (11%), Proteus vulgaris (9.0 %) and Brenneria nigrifluens (7.0%). Abo-Amer et al. (2008) claimed that infectious intestinal diseases were considered to be the major cause of hospitalization in almost all regions in Egypt. Microbiological examination of untreated water (hand pumps) samples revealed slightly contamination by faecal coliforms, faecal enterococci, Clostridium perfringens, Salmonella and Shigella.
During an experimental trial on captive stray dogs, after 3 months contact with these dogs, their attendants complained with gastrointestinal disturbances with or without fever. These captive dogs are incriminated as the source of this problem. The present study was carried out to evaluate the possibility that these dogs may serve as potential reservoirs of some zoonotic bacteria.
MATERIALS AND METHODS
The present study was carried out during the period from October to December, 2007. Identified blood and fresh faecal samples were collected from the target attendants and dogs. Samples were transferred for examination at the Department of Zoonoses, College of Veterinary Medicine, Cairo University, Egypt.
Humans
Two groups of male animal workers were investigated. First group of five animal attendants who are responsible for feeding, cleaning and collecting samples from dogs captive in boxes and the second group consists of 10 general animal workers and assistants in the same area of the study.
Dogs
Stray dogs (12 adult females and 15 puppies of 3-5 months old) were kept along the period of study captive in local private isolating pens (a concreted floor 1.5 m) (length), 1.0 (width) and 2.0 m (height) with wire door) at Abo-Rawash locality, El-Giza, Egypt. The animals were allocated as four adults, or five puppies per each pen. Dogs were fed ready prepared dog food supplemented with cooked chicken bone, while puppies were fed bread soaked in milk as well as cooked chicken bone.
Bacteriological Examination of Stool and Faecal Samples
Swab from each faecal sample was immersed directly into nutrient broth, tetrathionate broth and then incubated for 18-24 h at 37°C. Loopfuls from the broth were streaked onto blood agar, MacConkey agar, Xylose-Lysin Desoxycholate (XLD) agar, Eosine Methylen Blue (EMB) plates and Muller Hinton agar in microaerophilic atmosphere (Abdel-Aty and Rabie, 2003). The plates were Incubated at 37°C for 24-48 h. The suspected colonies were picked up, purified on trypticase soya agar and re-incubated at 37°C for another 24-48 h. Identification of the purified colonies were done according to the differences in morphology, cultural and biochemical characters (Koneman et al., 1992; Quinn et al., 1994).
Serological Diagnosis
Identification of Campylobacter
Using ACCUPROBER Campylobacter culture identification test kit (GEN-PROBE INCORPORATE, Sandigo, CA 92121), the tested faecal samples were initially cultivated using Campy Thioglycollate medium for 16-24 h at 37°C. Fifty microliter from the turbid portion of the broth added to the Prob Reagent tube and the test was run according to the kits manual for identification of 3 strains of Campylobacter (C. jejuni, C. coli and C. lari) using the control and reference samples associated with these kits.
Identification of Salmonella and E. coli
The isolated Salmonellae were serotyped according to Kauffmann-White Scheme (Kauffmann,1972) using polyvalent and monovalent sera of Salmonella 0 and H sera (Wellcome Reagents Limited, England). The suspected E.coli colonies were biochemically identified using API 20 diagnostic strip (Bio Merieux, France) and strains were identified serologically by commercial slide agglutination kit using polyvalent and monovalent somatic (0) and flagellar (H) antigens (Difco Laboratories USA) according to Ewing (1986).
Antimicrobial Susceptibility Testing
The disk diffusion method (Putnam et al., 2003) was used for susceptibility testing. Eight drugs were routinely used to test Gram negative enteric bacteria: Gentamycin (10 μg), Ampicillin (10 μg), Amoxicilin (20 μg), Nitrofurantoin (300 μg) Streptomycin (10 μg), Kanamycin (30 μg), Tetracyclin (30 μg) and cephalothin (30 μg).
RESULTS
In the current study bacteriological examination of stool samples collected from 15 dog’s attendants, (Table 1) revealed isolation of Enteroinvasive E. coli (EIEC) from 7 human (46.6%), Cambylobacter from two (13.3%) and Sallmonella from three (20.0%) attendants. The study cleared that E. coli infection was associated with diarrhea in four of direct contact workers and only two of other ten non contact workers. Meanwhile, Salmonella and Campylobacter infection were recorded in diarrheic close contact workers only.
On the other hand investigation of captive dogs (Table 2), evidenced infection by E. coli, 46.66 and 58.33% in puppies and adult dogs, respectively. Infection by Salmonella was 33.3 and 41.66% in both groups, respectively and finally infection by Campylobacter was 33.3% in both groups of dogs.
Concerning serotyping of the diagnosed isolates, (Table 3), thirteen Salmonella sub-typed were obtained. This include, S. typhimurium (2 from attendants and 6 from dogs), S. enteritides (one isolate) from human, moreover another (4) as untyped isolates from dogs. Serotyping of eleven Campylobacter isolates revealed detection of C. jejuni (2 from attendants and 4 from dogs), C. coli (3 isolates from dogs) and two untyped species were recorded from dogs also, (Table 4). Twenty one E. coli isolates were affiliated to five sero-groups as O 26 (5), O 76 (3) and O 55 (4) were isolated from both of attendants and dogs as in Table 5.
| Table 1: | Distribution of different bacteria in the examined attendants |
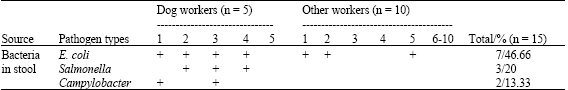 | |
| n = No. of examined samples | |
| Table 2: | Different bacterial pathogens diagnosed in the examined dogs |
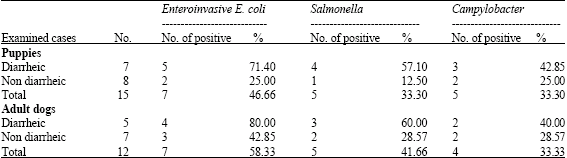 | |
| Table 3: | Serological identification of the isolated Salmonella |
 | |
| Table 4: | Serological identification of the isolated Campylobacter |
 | |
| Table 5: | Serological identification of the isolated Enteroinvasive E. coli |
 | |
| Table 6: | Antibiotic sensitivity of the isolated bacteria from human and dogs |
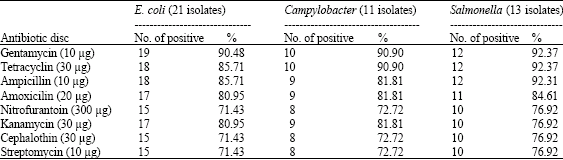 | |
Another serotypes as O 5 (2), O 111 (3) and four un-typed isolates were identified from dogs only as described in Table 5.
Sensitivity of different isolates was tested versus 8 antibiotic enriched disks (Table 6). The tested isolates showing high sensitivity for each of Gentamycine (10 μg) and Tetracycline (30 μg). Sensitivity percentage for the tested E. coli (21 isolates) was reached to 90.48 and 85.71%, for Campylobacter (11 isolates) was 90.9 and 90.9%, while it was 92.37 and 92.37% for Salmonella (13 isolates) versus the two previous mentioned antibiotic, respectively.
DISCUSSION
Zoonotic organisms such as viruses, bacteria or parasites can possess the potential role to cause severe diseases in both humans and animals. Free-ranging animals with sporadic or indirect contact to domestic livestock and humans may serve as reservoirs or sentinels for diseases (Aschfalk and Holler, 2006). So that continuous contact between diseased or carrier dogs and their workers under non proper hygienic measures initiate development of endemic foci for spreading of different pathogens, specially zoonotic one that have direct life cycle (Pullola et al., 2006).
In the present study bacteriological examination of workers stool samples revealed isolation of Enteroinvasive E. coli (EIEC) from human (46.6%), Cambylobacter (13.3%) and Sallmonella (20.0%). E. coli infection was associated with diarrhea. Diarrhea was observed in most of the direct contact workers and in a few of the non contact one. Meanwhile, Salmonella and Campylobacter infection were detected in diarrheic close contact workers only. These findings support the suggestion that the diagnosed symptoms in morbid cases consider as infection by these bacteria (Salmonella, Campylobacter and EIEC). This was agreed with Batt (2002) as these pathogens have been clearly associated with acute and chronic disease, typically causing diarrhea combined with tenesmus, vomiting, in-appetence, malaise, lethargy and abdominal pain are more variably encountered. These findings were at variance to those of Wierzba et al. (2006), who claimed that E. coli infection was not associated with diarrhea. This discrepancy could be related to the level of infection as well as the serotype of the diagnosed E. coli.
Concerning distribution of these pathogens in the examined dogs, infection by E. coli was ranged between 46.66 to 58.33%, by Salmonella was 33.3 to 41.66% and by Campylobacter was 33.3% in puppies and adult dogs, respectively. The infection was associated with diarrhea in 3 puppies only. These results were similar to those described by Hall and Simpson (2000) as Salmonella sp., Campylobacter sp. and EIEC were the main enteropathogens that most commonly identified in dogs. The isolated pathogens could be considered as the direct cause of the recorded diarrhea as previously mentioned by Brook (2006), but appearance or disappearance of these diarrhea may be affected by severity of infection, age as well as the general health conditions of the affected dogs. Brook (2006) asserted that there was relatively little information about the prevalence of pathogenic E. coli in dogs, but there was good evidence that they might play a role in the pathogenesis for both acute and chronic diarrhea. While Campylobacter sp. causing diarrhea in dogs, cats and humans especially in young. Moreover, the adult animals commonly have Campylobacter organisms living in their intestines but they do not experience any sickness due to it, while dogs can infect people whether they have diarrhea or not. In the present study, captive conditions of these dogs can act as stress factor led to occurrence of this diarrhea, this was agreed with Batt (2002). It has been estimated that the isolation of the same pathogens from the contact human evidenced that they are mainly originated from these dogs. This finding came in agreement with that of Hall (2004).
Concerning serotyping of the diagnosed Salmonella isolates, S. typhimurium were isolated from attendants and dogs, S. enteritides were recorded in attendants, while 4 untyped isolates were diagnosed from dogs. Campylobacter isolates include C. jejuni which diagnosed from attendants and dogs, while isolates of C. coli and two untyped species were diagnosed fom dogs only. Concerning the distribution of E. coli isolates, the serotypes of O 26 , O 76 and O 55 were identified from attendants and dogs while O 5 , O 111 and four un-typed isolates were diagnosed from dogs only. Diagnoses of the same serotype from attendants and their contact dogs explained the level of cross transmission between both of them. S. typhimurium and S. enteritides are known as more common in animals as well as causing human food poisoning. Most human cases of Salmonella infection cause fever, diarrhea and cramping that go away on their own. The disease is more severe in children, puppies and kittens (Brook, 2006). Likewise, C. jejuni is a major cause of infectious enteritis in human (Abdel-Aty and Rabie, 2003) and exposure to dogs with diarrhea will be triples a person's risk for developing enteritis from C. jejuni or C. coli (Brook, 2006). The number of bacterial genera isolated from the infected attendants and dogs in the present study considered to be few in comparison with that diagnosed in water by Abo-Amer et al. (2008) and Fareed et al. (2008) or that isolated from diarrheic children in Egypt by Wierzba et al. (2006). This difference explained that infection in the present study could be originate from local contamination for boxes of these dogs, as if this infection is originated from water supply, the problem can be recorded in peoples other than dog attendants.
It should be noted that persistence of infection around dogs was affected by survival time of each pathogen. Campylobacter can survive for days in surface water and as long as 4 weeks in feces; in addition, the duration of excretion in infected dogs can be as long as 4 months (Hall, 2004). Salmonella organisms are very difficult to remove from the environment, it easily to survive for 3 months in aquatic environments and in soil, shedding may continue for 3 to 6 weeks and can be reactivated at a later date by any inter-current stress, (Brook, 2006).
The tested isolates showing sensitivity for each of Gentamycine (10 μg) and Tetracycline (30 μg) enriched disks. According to Thompson and Roberts (2001), treatment of animals considered to be one of the fastest ways for eradication of different pathogens, the study advised to treat the morbid animal cases by Gentamycine 5 mg kg-1 b.wt., orally for 5 consecutive days (Schering-Plough Animal Health USA) with intravenous fluid therapy especially for puppies..
It has been estimated that captivity condition act as stress factor depress the host resistance and increase their susceptibility to infection and spread new pathogens. In the authors opinion, captivating of dogs under condition of the present study initiate development of endemic foci, as the infective stages were disseminated from infected dogs, in suitable conditions of dust, shad and moisture, these led to its propagation and persistence in the nature especially in and around the animal boxes so do potential zoonotic risks.
The study concluded that, captive dogs waiting for experimental studies must be thoroughly examined for all suspected pathogens, kept under strict hygienic measures and still under continuous observation along they remained in contact to human. Animal workers must be educated about transmission of different diseases. Moreover, they must be trained about how can they protect themselves from infection? In animal boxes, cleaning and disinfecting methods must be regularly applied inside and outside these boxes and completely separated from the surrounding area as well as it should be protect from rodents and vectors.
ACKNOWLEDGMENTS
The author expresses great appreciation for Prof. Dr. El-Seedy.F.R. Prof. of Microbiology and Vice-Dean of College of Veterinary Medicine, Beni-Suef University for his great effort revising this study.
REFERENCES
- Fareed, M.F., A.M. Haroon and S.A. Rabeh, 2008. Antimicrobial activity of some macrophytes from Lake Manzalah (Egypt). Pak. J. Biol. Sci., 11: 2454-2463.
CrossRefPubMedDirect Link - Aschfalk, N.A.K. and C. Holler, 2006. Campylobacter spp., Enterococcus spp., Escherichia coli. Salmonella spp., Yersinia spp. and Cryptosporidium oocysts in semi-domesticated reindeer (Rangifer tarandus tarandus) in Northern Finland and Norway. Acta Vet. Scandinavica, 48: 7-7.
CrossRefDirect Link - Pullola, T., J. Vierimaa, S. Saari, A.M. Virtala, S. Nikander and A. Sukura, 2006. Canine intestinal helminthes in Finland: Prevalence, risk factors and endoparasite control practices. Vet. Parasitol., 140: 321-326.
CrossRefDirect Link - Thompson, A.R. and M.G. Roberts, 2001. Does pet helminth prophylaxis increase the rate of selection of drug resistance?. Trends Parasitol., 17: 576-578.
PubMed - Wierzba, T.F., L.A. Abdel-Messih, R. Abu-Elyazeed, S.D. Putnam and K.A. Kamal et al., 2006. Clinic-based surveillance for bacterial- and rotavirus-associated diarrhea in Egyptian children. Am. J. Trop. Med. Hyg., 74: 148-153.
PubMed - Hall, E.J., 2004. Bacterial enteropathogens in dogs. Proceedings of the 29th World Congress of the World Small Animal Veterinary Association, Rhodes, Greece, Oct. 6-9, Langford Bristol, England, pp: 1-5.
Direct Link








