Research Article
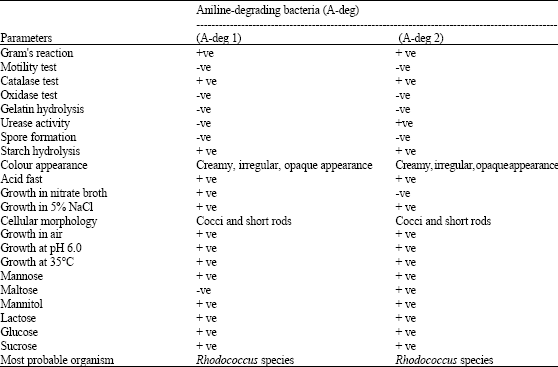
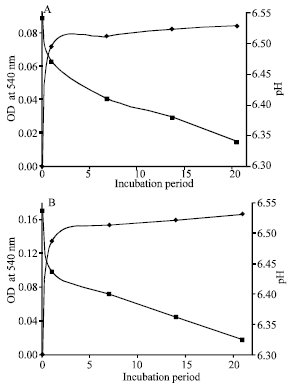
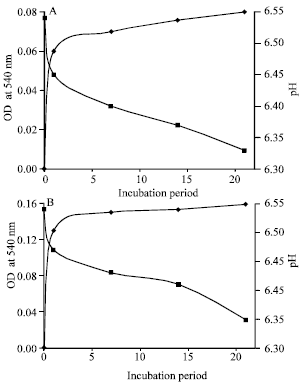
Biodegradation Potential of Two Rhodococcus Strains Capable of Utilizing Aniline as Carbon Source in a Tropical Ecosystem
1Department of Biological Sciences, College of Science and Technology, Covenant University, KM 10 Idiroko Road, Canaan Land, P.M.B. 1023, Ota, Ogun State, Nigeria
Nwodo-Chinedu Shalom
1Department of Biological Sciences, College of Science and Technology, Covenant University, KM 10 Idiroko Road, Canaan Land, P.M.B. 1023, Ota, Ogun State, Nigeria
O. Amund Olukayode
2Department of Botany and Microbiology, Faculty of Science, University of Lagos, Akoka-Yaba, Lagos, Lagos State, Nigeria