Research Article
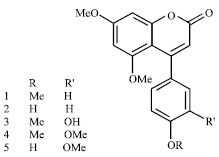
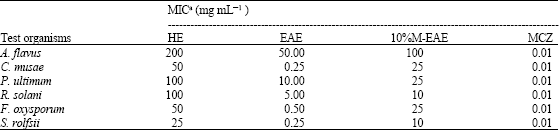
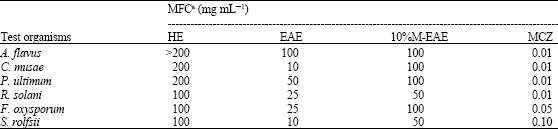
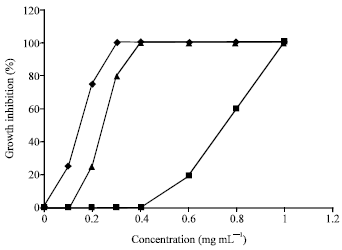
Synergistic Activities of 4-Arylcoumarins Against Phytopathogenic Fungi
Department of Microbiology, Faculty of Science, Silpakorn University, Nakorn Pathom 73000, Thailand
Asawin Wanbanjob
Department of Chemistry, Faculty of Science, Silpakorn University, Nakorn Pathom 73000, Thailand
Pittaya Tuntiwachwuttikul
Department of Chemistry, Faculty of Science, Silpakorn University, Nakorn Pathom 73000, Thailand
Yuemao Shen
Kunming Institute of Botany, The Chinese Academy of Sciences, Kunming 650204, China
Saisamorn Lumyong
Department of Biology, Faculty of Science, Chiang Mai University, Chiang Mai 50200, Thailand