Research Article
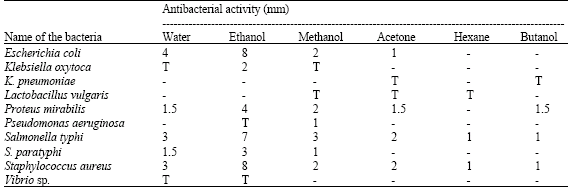
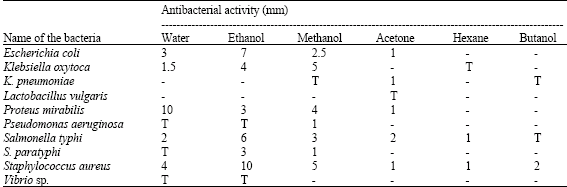
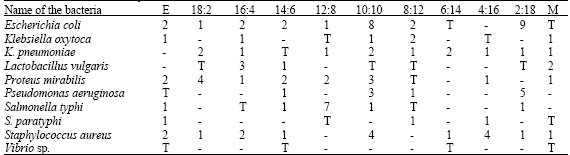
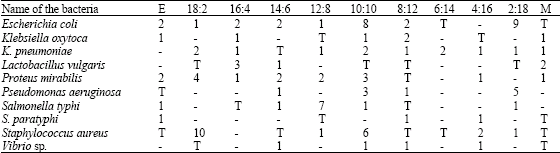
Antibacterial Activities of Green Mussel (Perna viridis) and Edible Oyster (Crassostrea madrasensis)
CAS in Marine Biology, Annamalai University, Parangipettai 608 502, Tamil Nadu, India
R. Anburaj
CAS in Marine Biology, Annamalai University, Parangipettai 608 502, Tamil Nadu, India
S. Jayalakshmi
CAS in Marine Biology, Annamalai University, Parangipettai 608 502, Tamil Nadu, India
R. Thavasi
CAS in Marine Biology, Annamalai University, Parangipettai 608 502, Tamil Nadu, India