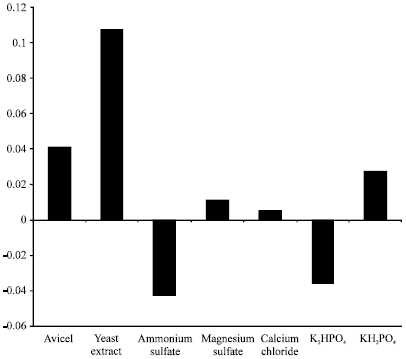
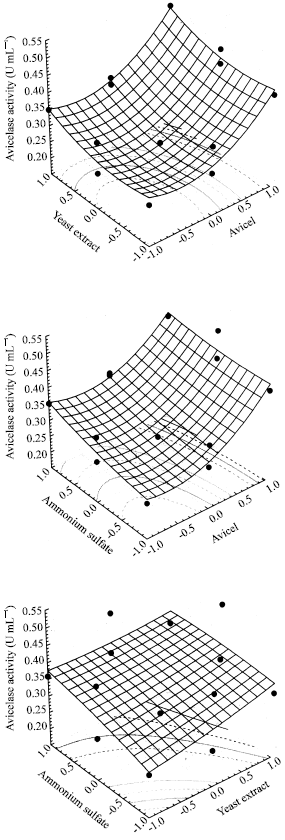
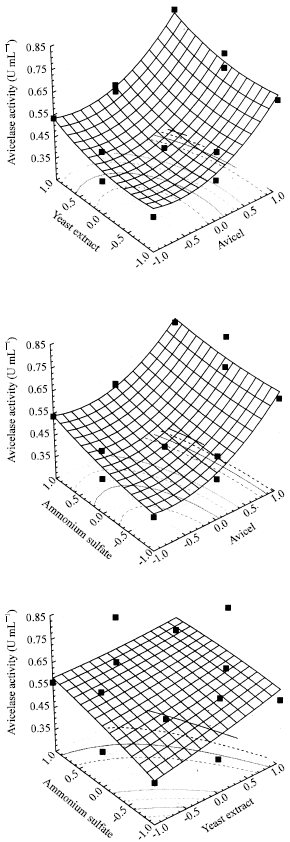
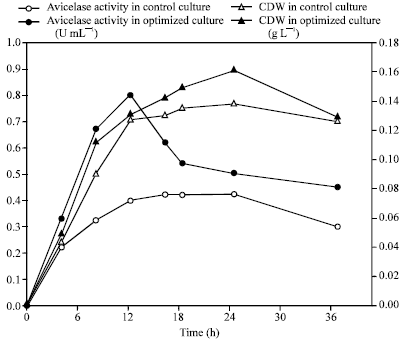
Research Article
Application of Factorial Designs for Optimization of Avicelase Production by a Thermophilic Geobacillus Isolate
Department of Bioprocess Development, Genetic Engineering and Biotechnology Research Institute, Mubarak City for Scientific Research and Technology Applications, New Burg El-Arab City 21934, Alexaudria, Egypt
Ehab R. El-Helow
Department of Botany, Faculty of Science, Alexandria University, Egypt
Khaled M. Ghanem
Department of Botany, Faculty of Science, Alexandria University, Egypt
Walid A. Lotfy
Department of Botany, Faculty of Science, Alexandria University, Egypt