Research Article
Composition and Antibacterial Activities of Tetrapleura tetraptera Taub. Pod Extracts
Department of Microbiology Michael Okpara University of Agriculture Umudike, Abia State, Nigeria
It has long been recognized that some plant materials exhibit antimicrobial properties. The use of these plant materials as preservatives and as means of preventing microorganism development in foods has become the subject of extensive studies (Gould, 1996). In particular, the inhibitory effects of extracts of many kinds of herbs and spices against food borne spoilage bacteria and pathogens have been reported. Among these are cassia, clove, garlic, sage, oregano, pimento, thyme and allspice (Shelef, 1983; Zaika and Kissinger, 1981; Salem and AiDelaimy, 1982; Tassou et al., 2000).
Currently, there is growing demand worldwide of consumers for minimizing chemical preservation that can be detrimental to human health (Cho et al., 1995; Smid and Gorris, 1999). Consequently spices, herbs and naturally occurring phenolics from various plants sources are being studied in detail in response to consumer requirements for fresher and more natural additive-free products (Nychas, 1995; Tassou et al., 1997).
Tetrapleura tetraptera Taub, family Mimosaceae, locally known as oshosho in South eastern Nigeria has widely varied applications in Nigerian folk medicine. The pods notably have an appealing culiniary use. Apparently, they are used to prepare soups for mothers from the first day of delivery to relieve post parturition contraction and as a lactation aid (Enwere, 1998). The antimicrobial activity of this plant has been exploited in the formulation of the dried powdered fruits of the plant. Thus dried powdered herbs have been formulated into soap bases using palm kernel oil (Adebayo et al., 2000). At the same time most of the folkloric claims agree in the traditional use of the fruit for management of convulsion, leprosy, inflammation and rheumatoid pains (Dalziel, 1948).
The molluscicidal activity of the extracts from the leaf, leaf stalk, stem-bark, root-bark have been exploited for long, but studies on the antibacterial effects of the essential oil from its fruits are scarce. Given the limited research information in this area the purpose of this study was to examine the antibacterial effects of the essential oil of the pods of T. tetraptera extracted using different solvents, to identify the chemical components of the extract and to determine at which concentration they were bacteriostatic and bactericidal to some food borne pathogenic bacteria. Such studies are essential if the full potential of T. tetraptera as a pharmacologic preparation in increasing the shelf-life of foods is to be exploited.
Sample Preparation
Pods of Tetrapleura tetraptera were obtained from commercial sources in Idah, Nigeria. The pods appeared to be sun-dried and were stored in air-tight containers until required for use. The pods were cut into small sized pieces before grinding in a coarse mill and finally into a powder with a Moulinex electric blender.
Extraction of Bioactive Materials
Fifty grams of milled pods was extracted in 250 mL of distilled water for 72 h at room temperature to obtain the aqueous extract, while another 50 g of the pod powder was extracted in 70% ethanol for 72 h to obtain the ethanolic extract. Both extracts were then filtered on Whatman No. 1 filter paper. The solvents were evaporated on an evaporator to obtain dry extracts. The dry solid was re-suspended in dimethyl sulfoxide (DMSO) to prepare various concentrations of both the aqueous and ethanolic extracts, which were used in the biological assay.
Bacterial Strains
The bacterial strains used in this study were as follows: E. coli (ATCC11775), P. aeruginosa (ATCC10145), Bacillus subtilis (ATCC6051) and Staphylococcus aureus (ATCC12600) obtained from Bioresources Development and Conservation Project (BDCP) of the University of Nigeria, Nsukka.
Stock cultures of the bacteria were routinely maintained on Mueller-Hintoon agar (Difco Laboratories, Detroit, USA) or broth for antimicrobial activity test. Trytone soy agar (Merck, Darmstadt Germany) was used to investigate the effects of extracts on the growth of the bacterial cells. All test strains were purified on MHA and identity confirmed by standard bacteriological methods (Collins and Lyne, 1984).
Phytochemical Analysis and Identification of Constituents
Extract components was subjected to standard phytochemical analyses for different constituents (Evans, 1983; Harbone, 1984). The presence of alkaloids, glycosides, tannins saponins and anthraquinones were tested. Structural determinations of the essential oils were based on special analysis: H-NMR spectra were recorded with a Varian EM300L spectrometer (Palo Ato, Carlifornia) and chemical shifts were given in ppm and IR spectra on a Perkin-Elmer Model 1320 spectrophotmeter (Connecticut, USA).
Disc Diffusion Assay
A 16 h culture was diluted with sterile physiological saline solution (0.85% (w/v) sodium chloride) with reference to the McFarland standard. The standard was prepared by adding an aliquot of 0.1 mL of 1% barium chloride to 0.9 mL 1% H2SO4 to achieve an inoculum of approximately 106 cfu mL-I. A 5 mL portion of this inoculum was placed onto the surface of pre-dried Mueller-Hinton agar plates and allowed to remain in contact for 1 min. The impregnated paper disc method (Davidson and Parish, 1989) was used to determine the antibacterial activity of the extract. Sterile 8 mm filter paper discs were used to absorb 1.5 mg of extract samples resuspended in DMSO and placed on top of the agar plate. Equal amounts of DMSO and benzoic acid were used as negative and positive controls, respectively. After allowing 1 h at room temperature for the samples to diffuse across the surface, the plates were incubated at 37°C for 24 h. The inhibition zone was measured in millimeter and the assay was carried out three times.
Determination of Bacteriostatic Concentrations
The plant extract was further tested to determine the concentrations at which they were bacteriostatic using the broth dilution technique (Anonymous, 1994). In order to test concentrations from 62.5-2% (w/v) the media containing 2 mg mL-I of the extracts were serially diluted two fold each with the media to concentrations of 2000, 1000, 500, 250, 125 and 62.5 μg mL-1.
The inoculum was prepared using a 16 h culture of the test organisms adjusted by reference to the McFarland standard and further diluted with MHB to achieve approximately 106 cfu mL-1. Equal amounts of DMSO and benzoic acid were used as negative and positive controls, Colonies formed were directly counted after incubation at 37°C for 24 h. Three replicates of each assay were carried out and the experiment was performed twice.
The bacteriostatic concentration was determined as the lowest concentration at which the bacterial cells in at least five of the six replicates was fully inhibited (Smith-Palmer et al., 1998; Burt and Reinders, 2003).
Effect of T. tetraptera Extracts on Viable Counts of Test Organisms in Culture Media
Trypticase soy Broth containing various concentrations of the plant extract was inoculated with the appropriate test organism (Staph aureus, E. coli, B. subtilis or P. aeruginosa) samples (0.1 mL) of serial dilutions of culture broth were spread on fresh TSB plates for enumeration of microbial populations after incubation at 37°C for 24 h.
Identification of Antimicrobial Substance in T. tetraptera Fruits
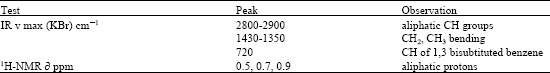
Preliminary phytochemical screening of T. tetraptera fruit pulp, shell and seed revealed that alkaloids, saponins, antraquinones were absent while tannins and glycosides were present (Table 1). Analysis of the ethanol extracts afforded active compounds which were characterized as tannins, cinnamic acids and carbohydrates by spectroscopic methods including IR and NMR. For example, a strong absorption band centered at between 2800 and 2900 cm-1 dominated the IR measurements. This absorption is linked to aliphatic C-H groups (C-H stretching) and indicates the presence of CH2 and CH3 groups (Table 2). The absorption bands in the region between 1430 to 1350 cm-1 are characteristics of CH2 and CH3 bending.
| Table 1: | Phytochemical screening of Tetrapleura tetraptera whole pod extracts |
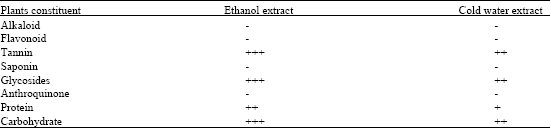 | |
| - Not detectable; + Low concentration; ++ Medium concentration; +++ High concentration | |
| Table 2: | Assignment of chemical shifts on the IR and 1H-NMR spectra of T. tetraptera pod extract |
 | |
| Table 3: | Antibacterial properties of T. tetraptera ethanolic extract against some food borne pathogens using the disc diffusion method |
 | |
| *Values are the averages of three replicates carried out twice | |
A band approximately at 720 cm-1 assigned to C-H groups may be characteristic of 1,3-bisubstituted benzene (Kemp, 1988). The 1H-NMR (CCI4) spectrum of the extract contained resonance signals at 0.5, 0.7 and 0.9 ppm, which corresponded to the protons of the functional groups of fatty acids sugars (-CH2-, CH3CO-). Careful evaluation of our analytical data as well as data reported in the literature suggests that the T. tetraptera essential oil extract contain oleanolic glycosides, tannins and cinnamic acids. These data are consistent with reports of the presence of tannins, oleanolic glycosides and cinnamic acids (Adesina et al., 1980) in T. tetraptera fruit pods.
These phytochemical metabolites detected in this study are effective inhibitors of the growth of yeasts, bacteria and molds as well as toxin production by microorganisms. Cinnamic acid has been reported to inhibit the growth of a number of bacteria such as Staphylococcus sp. Micrococcus sp. Bacillus sp. and Enterobacter sp. at 500 μg mL-1 (Masuda et al., 1998). It has been reported that application of 10 mg disc-1 of tannin methyl gallate produced moderate antimicrobial effects against C. perfriingens, S. aureus, E. coli and B. fragilis (Ahn et al., 1998). The concentrations at which the extract of T. tetraptera exerts it inhibitory effect indicate that it might be useful in the biopreservation of foodstuffs where the prevention of growth of spoilage bacteria is desired.
Antibacterial Activity of T. tetraptera Extract
The growth inhibitory activity of the ethanol extracts of T. tetraptera on some food borne bacteria is presented in Table 3. The responses varied with the bacterial strain tested. The extracts produced strong antibacterial effect on Staph aureus (ATCC12600), E. coli (ATCC11775) and P. aeruginosa (ATCC10145) whereas the growth of B. subtilis (ATCC6051) was weakly affected. A number of essential oil components has been identified as effective antibacterials. In vitro studies have demonstrated antibacterial activity of carvacrol, thymol, eugenol, cinnamaldehyde and cinnamic acid (Burt, 2004), against foodborne pathogens such as S. aureus, B. cereus, E. coli and L. monocytogenes.
Mic of the T. tetraptera Extract Against Various Foodborne Bacteria
The MIC of the plant extract was determined on the selected foodborne bacteria. E. coli (ATCC11775) P. aerugionsa (ATCC10145) and S. aureus (ATCC12600) showed higher sensitivity than B. subtilis (ATCC6051). The MIC of the substance was 250 μg mL-1 against E. coli, P. aeruginosa and S. aureus. Against B. substilis, the MIC of the plant extract (500 μg mL-1) was found to be higher than those of sorbic acid (250 μg mL-1 or benzoic acid (Table 4).
The test organisms used in this study are common food pathogens, which are associated with many serious foodborne illnesses. Recently, there appears to be growing concern about food safety, consumers desiring fewer synthetic additives and products with a smaller impact on the environment (Burt, 2004). There is therefore scope for new methods of safe antimicrobials, which have a natural image as an alternative of the chemical preservation in foods. IIt has long been recognized that most spices are more active against Gram-positive bacteria than Gram-negative bacteria (Masuda et al., 1998). However, the ethanolic extract of T. tetraptera was effective for E. coli and P. aeruginosa at MIC of 250 ug mL-1 as against the MIC of 500 mg mL-1 for B. subtilis.
Growth in Culture Media
The growth of B. subtilis was slightly delayed by 8% (w/v) of the extract of T. tetraptera with 2 log microbial reduction after 24 h. The extract however at 4% concentration, provided 2-6 log reductions for Staph aureus (ATCCC12600), P. aeruginosa (ATCC10145) and E. coli, (ATCC11775).
Many spices and plant essential oils possess antimicrobial properties (Zaika, 1988; Nychas, 1995; Burt, 2004). The results obtained from this study concurred with it (Table 5). Whereas the growth inhibition zones measured by disc diffusion show a strong antibacterial effect on the test organisms, the time kill performance in broth culture was, however less substantial. Furthermore, at concentrations of 8% of the extract, the number of viable cells decreased by approximately 2-6 log factors over a period of 24 h at 37°C but a total kill was apparently not achieved. Increasing the extract dose may completely inhibit the growth of the organisms.
In previous reports the extent of inhibition of spices and their extracts was affected by the concentrations applied. The addition of 8 or 10% of P. densiflora extract to culture broth completely inhibited the growth of B. subtilis Salmonella typhimurium and Staphyloccus aureus (Kim and Shin, 2004). In this study 8% of T. tetraptera extract had almost the same capacity inhibiting B. subtilis (ATCC6051) as that in 4% concentration for the rest of the test bacterial strains demonstrating that its resistance to T. tetraptera is high.
In general, the active antimicrobial compounds of spice essential oils are terpenes, which are phenolic in nature (Tainter and Grenis, 2001). It has been reported that phenolic compounds cause outer membrane disintegration and increased permeability of sensitive cells as antimicrobial mechanisms (Brul and Coote, 1999). In addition, the mode of action of these compounds is concentration dependent as was evident in this study.
| Table 4: | Comparison in the minimal inhibitory concentration of T. tetraptera extract and synthetic preservatives against various foodborne bacteria |
 | |
| Table 5: | Effect of T. tetraptera extract against several foodborne microorgamisms in broth culture (log10 cfu mL-1*) |
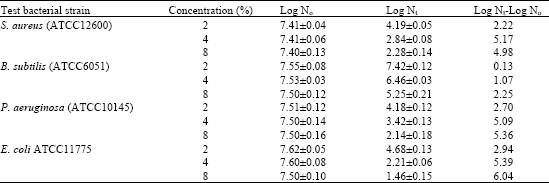 | |
| *Each value is the mean of triplicate samples taken from two different experiments±standard deviation | |
Naturally occurring substances in plants often play an important role in controlling the growth of spoilage and pathogenic microorganisms in foods (Burt, 2004). In this study, T. tetraptera extract produced a clear inhibitory effect on some potentially harmful bacteria. However, B. subtilis, a Gram-positive rod was slightly affected. Present results indicate that the growth-inhibitory effect was concentration dependent. It is well understood that antimicrobial effects observed in broth systems do not necessarily occur in complex foods. Further work is necessary to establish whether the activity of T. tetraptera is exerted in complex food systems.