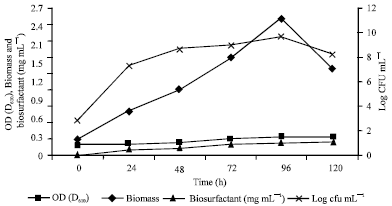
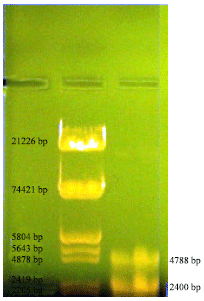
Research Article
Biodegradation of Crude Oil by Nitrogen Fixing Marine Bacteria Azotobacter chroococcum
CAS in Marine Biology, Annamalai University, Parangipettai-608 502, Tamil Nadu, India
S. Jayalakshmi
CAS in Marine Biology, Annamalai University, Parangipettai-608 502, Tamil Nadu, India
T. Balasubramanian
CAS in Marine Biology, Annamalai University, Parangipettai-608 502, Tamil Nadu, India
Ibrahim M. Banat
School of Biomedical Sciences, University of Ulster, Coleraine BT52 1SA, Northern Ireland, UK