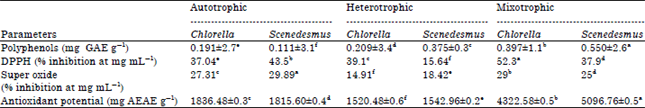
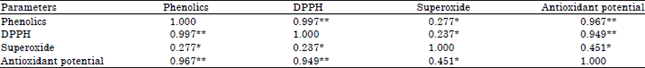
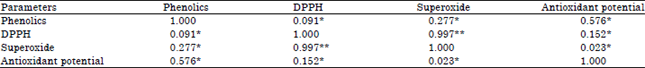
Research Article
Relationship Between Total Phenolics Content and Antioxidant Activities of Microalgae Under Autotrophic, Heterotrophic and Mixotrophic Growth
Department of Biotechnology, Indian Academy Degree College Centre for Research and Post Graduate Studies, Bengaluru, 560043, Karnataka, India
G. Sibi
Department of Biotechnology, Indian Academy Degree College Centre for Research and Post Graduate Studies, Bengaluru, 560043, Karnataka, India