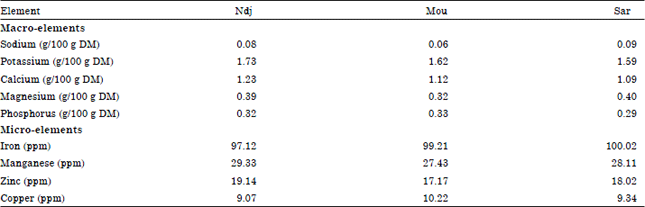
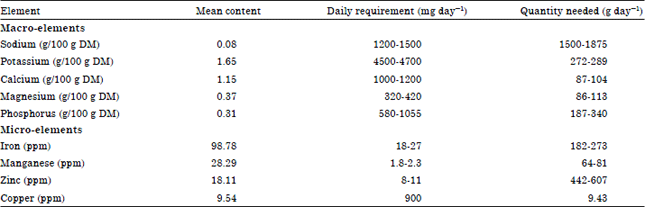
Research Article
Proximal and Elemental Composition of Moringa oleifera (Lam) Leaves from Three Regions of Chad
Research Laboratory of Natural Substances, Faculty of Applied and Exact Sciences, University of N�Djamena, P.O. Box 1027, Chad
Tarkodjiel Mianpereum
Research Laboratory of Natural Substances, Faculty of Applied and Exact Sciences, University of N�Djamena, P.O. Box 1027, Chad
Ngakou Albert
Department of Biological Sciences, University of Ngaoundere, P.O. Box 454, Ngaoundere, Cameroon