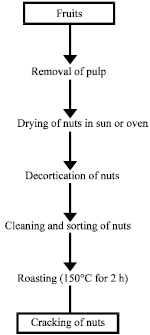
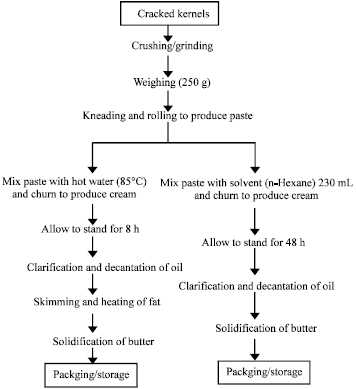
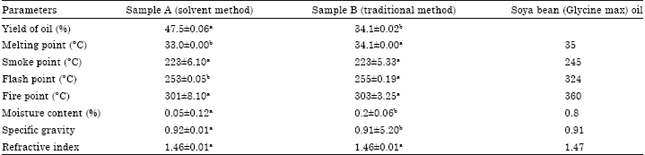
Research Article
Effects of Extraction Methods on the Yield and Quality Characteristics of Oils from Shea Nut
Department of Food Science and Technology, University of Agriculture, Makurdi, P.M.B. 2373, Makurdi, Benue State, Nigeria
Lilly N. Umenger
Department of Food Science and Technology, University of Agriculture, Makurdi, P.M.B. 2373, Makurdi, Benue State, Nigeria
Ayangealumun Iorbee
Department of Food Science and Technology, University of Agriculture, Makurdi, P.M.B. 2373, Makurdi, Benue State, Nigeria