Research Article
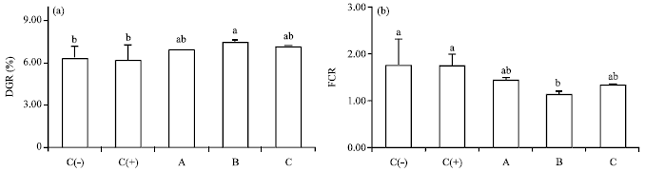
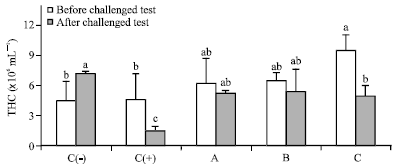
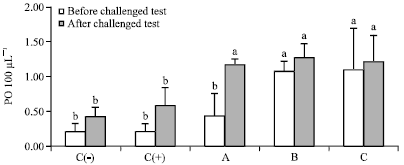
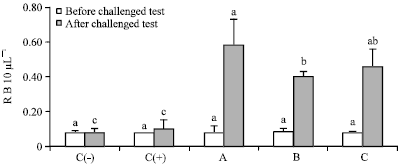
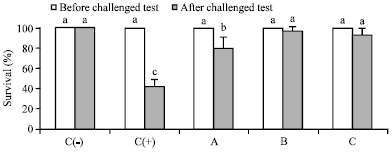
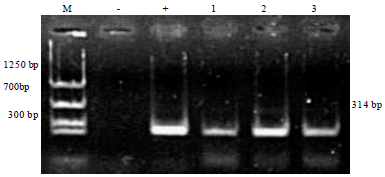
Dietary Synbiotic Influence on the Growth Performances and Immune Responses to Co-Infection with Infectious Myonecrosis Virus and Vibrio harveyi in Litopenaeus vannamei
Post Graduate School of Aquaculture Science, Bogor Agricultural University, Indonesia
Widanarni
Department of Aquaculture, Faculty of Fisheries and Marine Science, Bogor Agricultural University, Indonesia
Munti Yuhana
Department of Aquaculture, Faculty of Fisheries and Marine Science, Bogor Agricultural University, Indonesia