Research Article
Inclusion of Purple Non-sulfur Bacterial Biomass in Formulated Feed to Promote Growth, Feed Conversion Ratio and Survival of Asian Seabass Lates calcarifer Juveniles
Borneo Marine Research Institute, Universiti Malaysia Sabah, Jalan UMS, 88400 Kota Kinabalu, Sabah, Malaysia
Tan Ee Ting
Borneo Marine Research Institute, Universiti Malaysia Sabah, Jalan UMS, 88400 Kota Kinabalu, Sabah, Malaysia
Sujjat Al-Azad
Borneo Marine Research Institute, Universiti Malaysia Sabah, Jalan UMS, 88400 Kota Kinabalu, Sabah, Malaysia











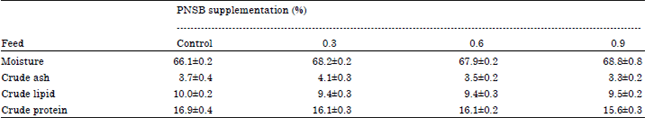
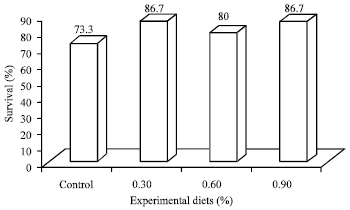
G.ABIRAMI Reply
i am vary interested with this article .kindly send me the full PDF for mete