Research Article
Trophic Level of Fishes Associated in the Trawl Bycatch from Parangipettai and Cuddalore, Southeast Coast of India
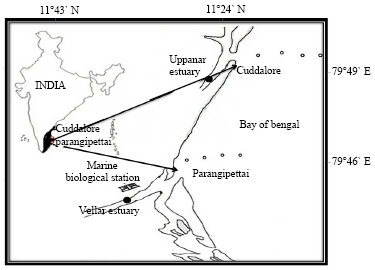
Centre of Advanced Study in Marine Biology, Faculty of Marine Sciences, Annamalai University, Parangipettai-608 502, Tamil Nadu, India
S. Purusothaman
Centre of Advanced Study in Marine Biology, Faculty of Marine Sciences, Annamalai University, Parangipettai-608 502, Tamil Nadu, India
S. Muthuvelu
Centre of Advanced Study in Marine Biology, Faculty of Marine Sciences, Annamalai University, Parangipettai-608 502, Tamil Nadu, India