Research Article
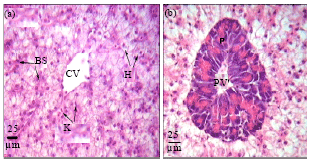
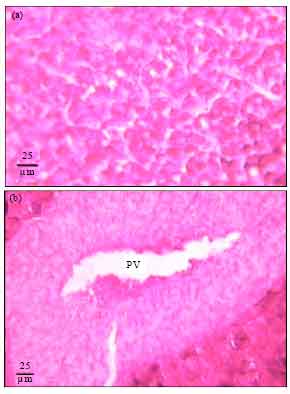
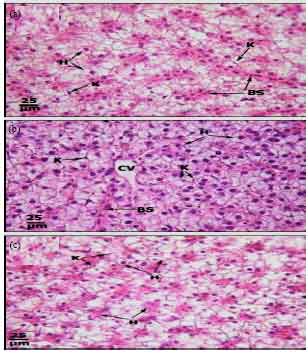
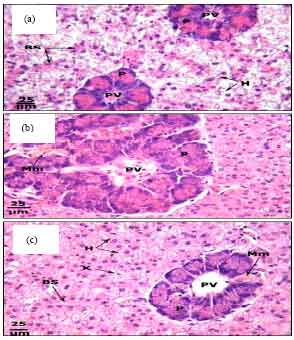
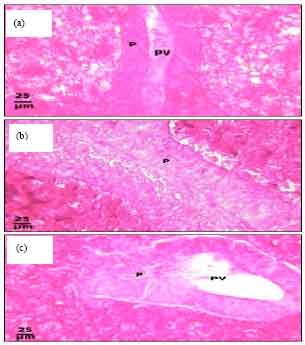
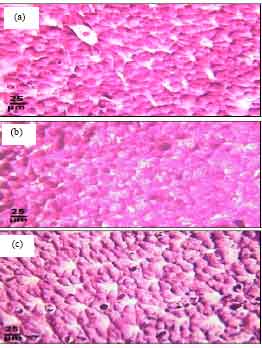
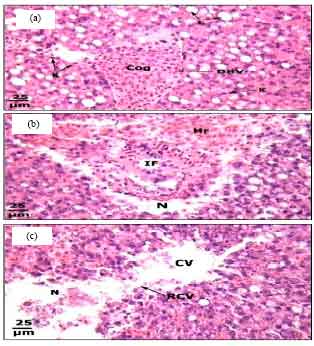
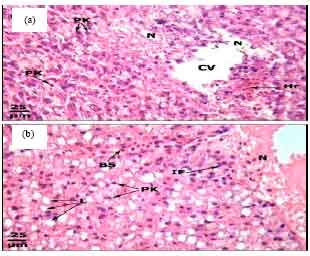
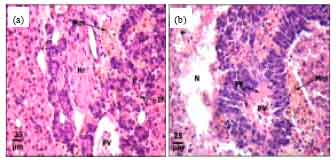
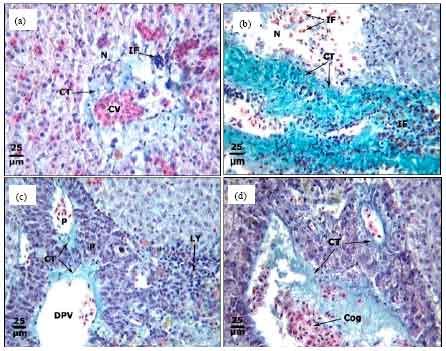
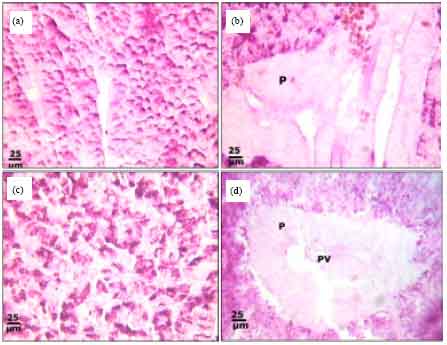
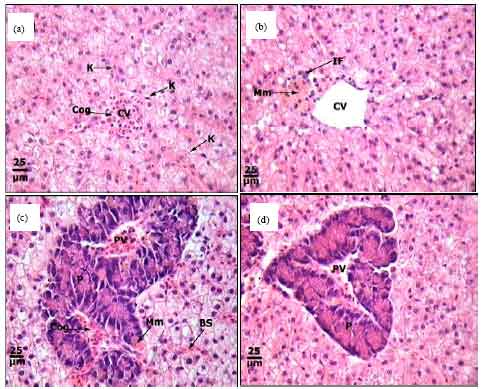
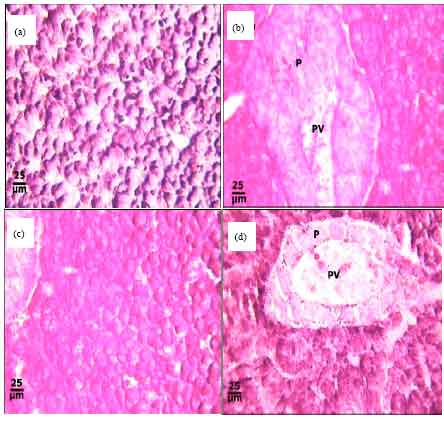
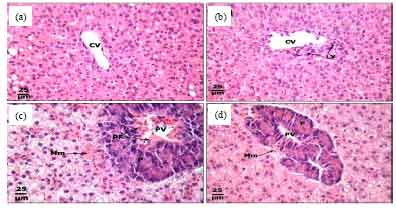
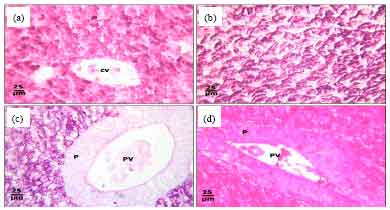
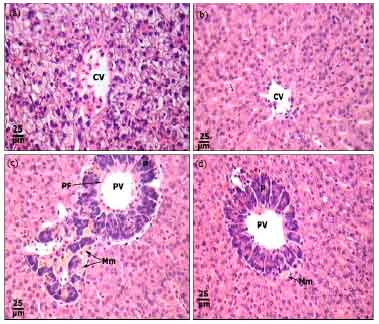
Protective Roles of Tomato Paste and Vitamin E on Cadmium-induced Histological and Histochemical Changes of Liver of Oreochromis niloticus (Linnaeus, 1758)
Department of Zoology, Faculty of Science, Assiut University, Egypt
U.M. Mahmoud
Department of Zoology, Faculty of Science, Assiut University, Egypt
E.T. Wassif
Department of Zoology, Faculty of Science, Assiut University, Egypt
M. Naguib
Department of Zoology, Faculty of Science, Assiut University, Egypt