Research Article
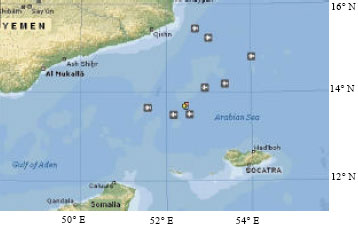
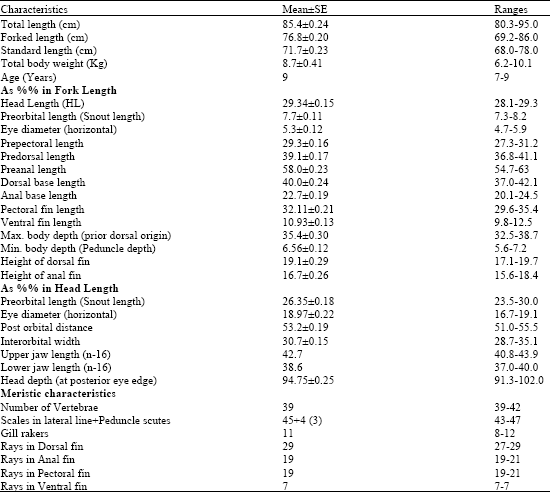
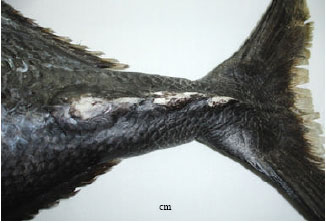
Additions to Benthopelagic Fish Fauna of the Aden Gulf-Arabian Sea(Actinopterygii: Bramidae and Sternoptychidae)
Faculty of Environmental Sciences and Marine Biology, Hadhramout University, Mukalla, Yemen
A.H. McNoon
Brum Fisheries Company, Shehir, Yemen