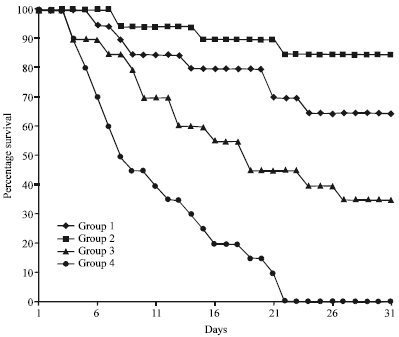
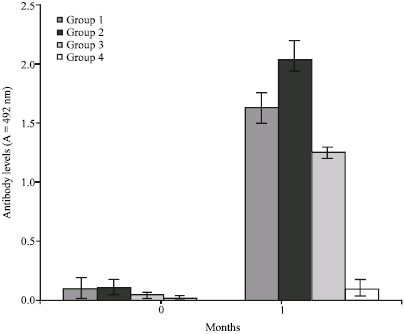
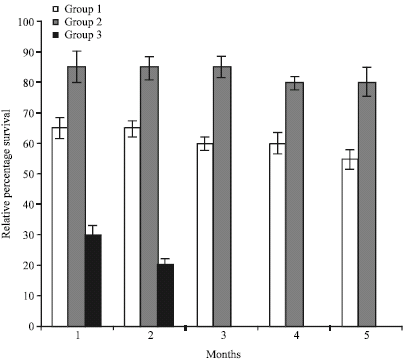
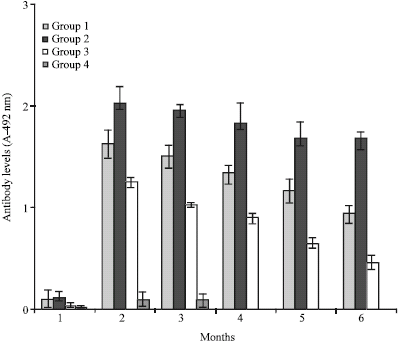
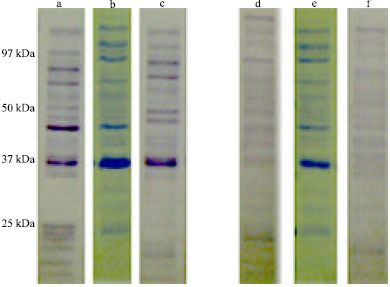
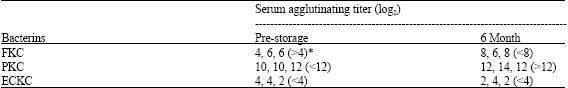
Research Article
Stability of Effective Edwardsiella tarda Vaccine Developed for Japanese Eel (Anguilla japonica)
Fish Disease Laboratory, Department of Aquaculture, Faculty of Agriculture, Kochi University, Nankoku Shi, Kochi 783-8502, Japan
Kenji Kawai
Fish Disease Laboratory, Department of Aquaculture, Faculty of Agriculture, Kochi University, Nankoku Shi, Kochi 783-8502, Japan