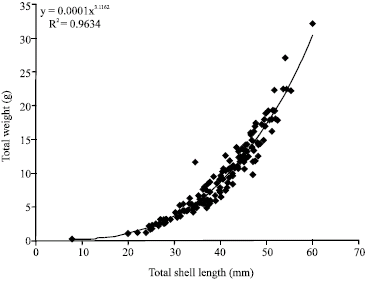
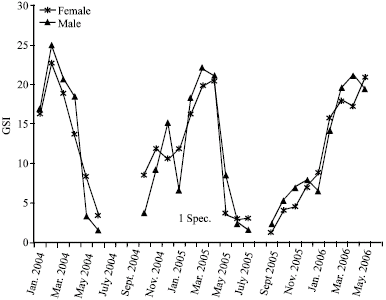
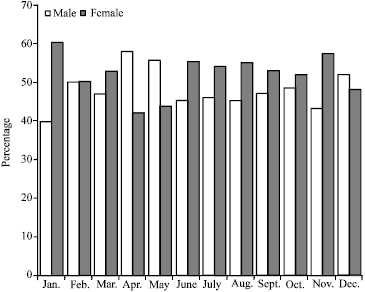
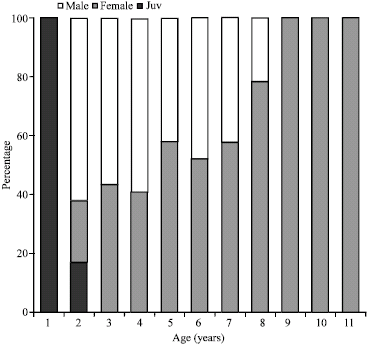
Research Article
First Remarks on Abalone Biology (Haliotis pustulata) on the Northern Coast of Aden Gulf, Yemen
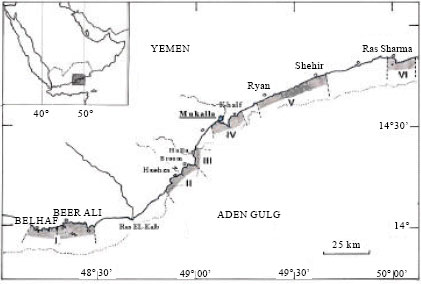
Faculty of Environmental Sciences and Marine Biology, Hadhramout University of Sciences and Technology, Mukalla, Yemen
A.A. Basmidi
Faculty of Environmental Sciences and Marine Biology, Hadhramout University of Sciences and Technology, Mukalla, Yemen
M.Sh. Aideed
Faculty of Environmental Sciences and Marine Biology, Hadhramout University of Sciences and Technology, Mukalla, Yemen
Al-Quffail A. Saeed
Faculty of Environmental Sciences and Marine Biology, Hadhramout University of Sciences and Technology, Mukalla, Yemen