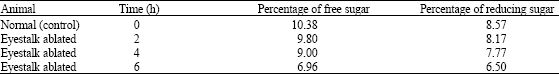
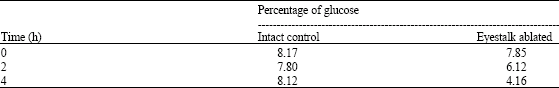
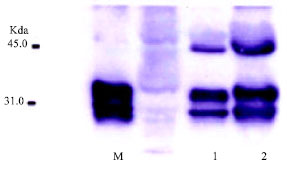
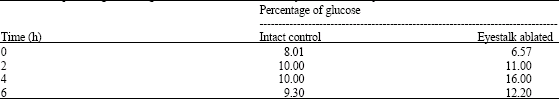
Research Article
Activity of Hyperglycemic Hormone in the Eyestalk of Commercially Important Crab Portunus pelagicus (Linnaeus)
Centre of Advanced Study in Marine Biology, Annamalai University, Parangipettai-608 502, Tamil Nadu, India
P. Soundarapandian N. John Samuel
Centre of Advanced Study in Marine Biology, Annamalai University, Parangipettai-608 502, Tamil Nadu, India
S. Rajagopal
Centre of Advanced Study in Marine Biology, Annamalai University, Parangipettai-608 502, Tamil Nadu, India