Research Article
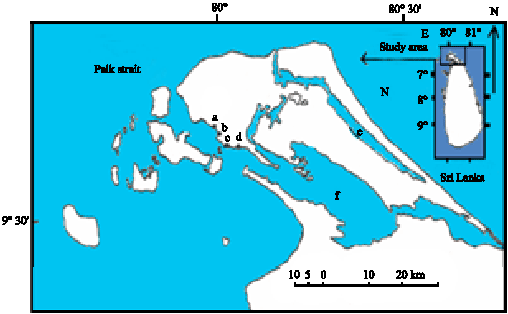
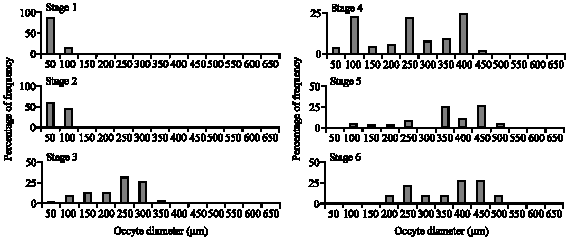
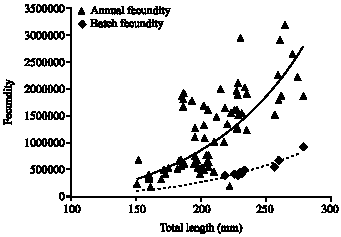
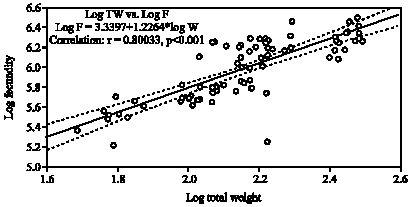
Fecundity Studies of Gerres abbreviatus (Bleeker, 1850) From the Jaffna Lagoon, Sri Lanka
Department of Zoology, University of Jaffna, Jaffna, Sri Lanka
G.A. Charles
Department of Zoology, University of Jaffna, Jaffna, Sri Lanka
S. Shutharshan
Department of Zoology, University of Jaffna, Jaffna, Sri Lanka