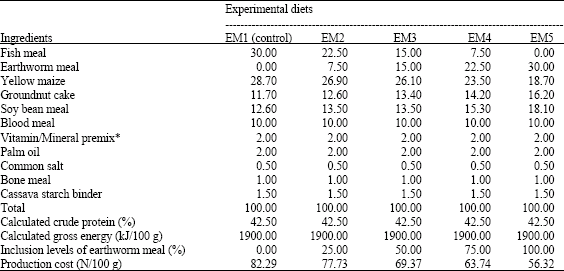
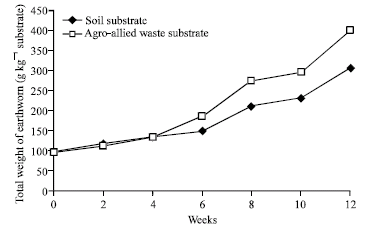
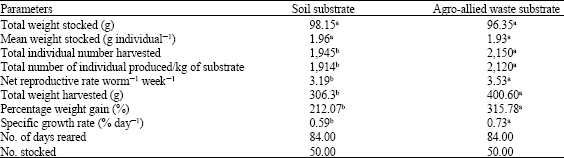
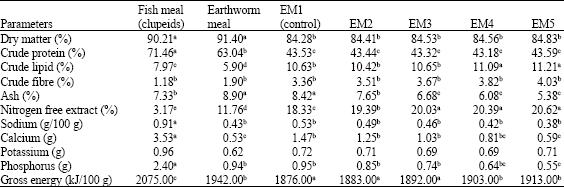
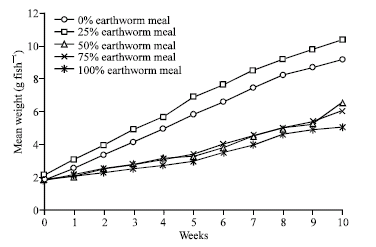
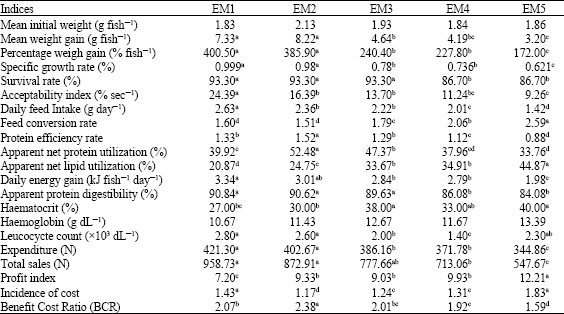
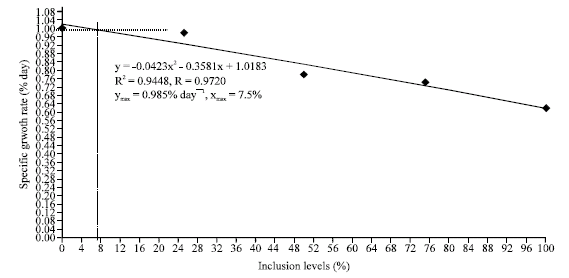
Research Article
Culture and Utilization of Earthworm as Animal Protein Supplement in the Diet of Heterobranchus longifilis Fingerlings
National Institute for Freshwater Fisheries Research, P.M.B. 6006, New-Bussa, Niger-State, Nigeria
A.A.A. Ugwumba
Department of Zoology, University of Ibadan, Ibadan, Nigeria
C.T. Madu
National Institute for Freshwater Fisheries Research, P.M.B. 6006, New-Bussa, Niger-State, Nigeria
S.S. Eze
National Institute for Freshwater Fisheries Research, P.M.B. 6006, New-Bussa, Niger-State, Nigeria
J. Isa
National Institute for Freshwater Fisheries Research, P.M.B. 6006, New-Bussa, Niger-State, Nigeria