ABSTRACT
African catfish larvae, Clarias anguillaris were reared on monospecies culture of the freshwater rotifer, Brachionus calyciflorus in tanks in comparison with a mixed zooplankton diet from the time the fry were 5 day old through to 24 days of age. Attempt was made to isolate the freshwater rotifer, Brachionus calyciflorus by chemical treatment using an organophosphorus acid ester diazinon whose concentration was determined in another reported investigation in the laboratory. At the beginning of feeding, larvae had a mean weight of 5.08±1.28 mg and standard length of 9.20±1.56 mm, respectively. Results obtained indicated that survival was relatively high in all treatments. Growth measured in terms of gain in weight and length increases were not significantly different among the treatments although higher in the fry fed rotifer enriched diet. The use of zooplankton cultures may circumvent problems associated with rotifer availability in fish ponds. The implication of the result to management and successful culture of the African catfish larvae are discussed.
PDF Abstract XML References Citation
How to cite this article
DOI: 10.3923/jfas.2007.275.284
URL: https://scialert.net/abstract/?doi=jfas.2007.275.284
INTRODUCTION
The African catfish, Clarias anguillaris is recognized as an ideal candidate for aquaculture due to its continued reproductive performance under intensive rearing culture conditions, ability to efficiently utilize commercial feed stuffs and the capacity to use atmospheric oxygen (Jeje, 1992). The African catfish generally are valued as one of the highest quality freshwater fish in Nigeria (Mohammed and Omoregie, 2004). Despite the cultivable characteristics of clariid catfish species, availability is limited due to an inadequate hatchery system and insufficient live food of good quality constitute a major problem to fish farmers and hatchery operators.
Culturing of the African catfish fry to fingerlings in tanks would allow monitoring of survival, growth and enable the culturist to maintain optimal temperature and other growing conditions (Arimoro, 2005). Also for commercial fingerling producers it would eliminate the need and the stress involved in harvesting fingerlings from ponds and transporting them to holding tanks before shipping to growers (Ludwig and Lochmann, 2000).
Successful culture of most fish fry requires the presence of the smallest zooplankton, mainly rotifers, as first food (Arimoro and Ofojekwu, 2003). It has also been shown that is still better for the first feeding larvae of most fish species since it leads to healthier larvae growth (Wang et al., 2005). These organisms must be able to meet their nutritional requirements for optimization of growth and survival (Shiri et al., 2003). According to Ajah (1998), the choice of rotifers for early fish larvae has the advantage of providing a low cost, highly predictable and reliable in the production of food organism which is acceptable and easily caught by the fish larvae. Several techniques have been adopted for the culture of the fresh water rotifer, Brachionus calyciflorus (Lubzens et al., 1989; Hoff and Snell, 1997). Rotifers have the additional advantage of being maintainable in a stable culture by feeding them recycled diets in a feed back culture system (Hirata and Yamasaki, 1983; Hirata et al., 1983).
Maintaining monocultures of the freshwater rotifers is complicated (Ludwig and Lochmann 2000). It requires setting up a rotifer culture and a microalgae culture to feed the rotifers. Both cultures require constant care, precise growing conditions, specialized equipment isolation to avoid contamination (Arimoro, 2006). Jeje (1992) reflected on the difficulty for establishing pure cultures of monospecific zooplankton and thus suggested that ‘for hatchery operations, especially in developing nations like Africa the development of a mixed culture of zooplankton population would be more realistic as the techniques involved can be more easily mastered by hatchery managers.
Several chemicals {trichlorfon (Dylox®), fenthion (Baytex) and diflubenzuron (Dimilin)} have been found effective for initiating a monoculture of rotifer in ponds (Burtle and Morrison, 1987; Ludwig, 1993). Most of these chemicals are organophosphates which known to be toxic to cladocerans, slightly toxic to free swimming copepods and their nauplii, but not fatal to rotifers at levels below 1.5 mg L-1, hence they can be used in initiating monocultures of rotifers.
There are considerable reports on feeding successes using the marine rotifer, Brachionus plicatilis as live food for several marine and few freshwater species (Ottera, 1993; Craig et al., 1994; Castell et al., 2003). Only a few documented studies are available on the use of the freshwater rotifer, B. calyciflorus for raising of freshwater fish species. For example, Lim and Wong (1997) reported rearing successes with the larvae of the ornamental discus raised on this rotifer Ludwig (1993) demonstrated that consistently high survival rates for the sunshine bass fry are possible when the fry are stocked just before predatory copepods are present.
Similarly, Shiri et al. (2003) and Arimoro and Ofojekwu (2003) reported over 60% survival rates with the freshwater fish larvae, burbot (Lota lota) and the African toothed carp (Aphyosemion gardneri) raised on this rotifer. The difficulty in raising most fish species from fry stage to fingerling still remains a global problem. Accordingly, this study provides useful information on the successful rearing of the African catfish, Clarias anguillaris larvae on the cultured freshwater rotifer, B. calyciflorus in comparison with a mixed zooplankton diet.
MATERIALS AND METHODS
This experiment was carried out in the fishery complex unit of the department of Zoology, University of Jos, Jos Nigeria.
Larvae were fed daily with each of the experimental diets to satiation point marked by cessation of feeding, non-wagging of caudal fin and resting/withdrawal stage by the larvae.
Culture of the Freshwater Rotifer, B. calyciflorus
The freshwater rotifer, B. calyciflorus was first isolated from a mixed population of zooplankton comprising rotifers, copepods and Cladocerans from a 300 L basin duck weed tank situated in the Zoological garden of the University of Jos, Jos Nigeria. Pure cultures were obtained by a series of chemical treatments with diazinon (Basudine®) to establish the safe concentration for rotifers. This was achieved through another published investigation in the same laboratory by Agbon et al. (2002) as 1.2 mg L-1 of the active ingredient. At this concentration, Crustaceans (Cladocerans and copepods), aquatic insects including mosquito larvae failed to survive thereby allowing the rotifers to multiple. A more detailed method of culture is contained in Arimoro and Ofojekwu (2004) and Arimoro (2005). This rotifer was mass produced in 30 L plastic tanks using algae species Scenedesmus sp. and Chlorella sp. in combination with baker’s yeast. In addition, a modified method for continuous culture of the rotifers was employed (Hirata and Yamasaki, 1983). In modifying the previous method an organophosphorus acid ester (diazinon) applied at the rate of 1.2 mg L-I, was used to obtain a pure culture of rotifers. Mixed algal species particularly, Scenedesmus sp. and Chlorella sp. were used in the continuous culture of the rotifers instead of the unialgal culture used in the previous method. Another important modification was that rotifers were siphoned from the top of the aliquot without disturbing the bottom, instead of filtering as was the case in the original method (Fig. 1). Culture sediments such as faeces and excess food were fermented in a bucket for 1-2 weeks and to this was added 50 μg L-1 of vitamin B12. The vitamin favored the growth of bacteria for biodecomposition of the sediments. The resulting fermented biomass was then used for the cultivation of the mixed algae.
The algal produced from these subsistence nutrients were fed back to the rotifers in the form of recycled diets. In this way, continuous, steady cultures of algae and rotifers were maintained. This was continued for the whole length of the experiments.
Enrichment Medium
The freshwater rotifers, B. calyciflorus used in these experiments were divided into two groups. The first group was enriched in cod liver oil before harvesting for feeding the larvae. 3.5 mL of cod liver oil was applied per 20 L of rotifer culture. This was continued for 12 h every night while keeping an eye on the water quality through the period (Castell et al., 2003; Craig et al., 1994). The second groups of rotifers were not enriched in the cod liver oil.
Culture of Mixed Zooplankton
For culture of mixed zooplankton no chemical was used. The initial cultures from the wild were fed with algal diets and this ensured their replication especially the cladocerans and the copepods. The rotifers were reduced in these cultures probably due to the fact that they were preyed upon by the larger zooplankton.
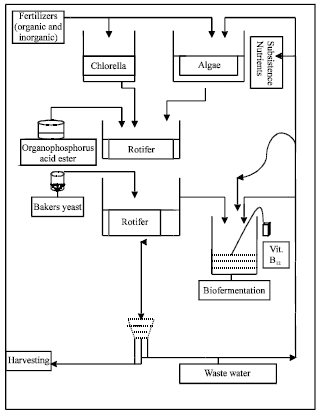 | |
| Fig. 1: | Continuous culture of rotifers in a feed back mechanism |
Harvesting of Zooplankton and Feeding of the African Catfish Larvae
Diet 1 (rotifer unenriched) and Diet 2 (enriched rotifers) were harvested periodically with a 50 μm size sieve and fed to the African catfish larvae daily. Rotifer density in the larval culture tanks was maintained at over 30 individuals per mL. Rotifer concentrations were determined with a sedge wick-rafter cell and a microscope.
Diet 3 Mixed Zooplankton comprising copepods, rotifer and cladocerans were harvested from their culture tanks using a 100 μm size sieve.
Clarias anguillaris fry (mean weight and length of 5.08±1.28 mg and 9.20±2.56 mm, respectively) were obtained from the homogenous stock produced in the indoor hatchery by hormone induced breeding in the Rock waters fish farm in Jos. They were assigned randomly to two replicates glass tanks (60x30x30 cm) for each of the three treatments with aeration and a static water system filled with 40 L of dechlorinated tap water. Water was partially replaced after two weeks of larval culture. One hundred fry were introduced initially into every fry tank.
Larval Measurements
Catfish fry of initial mean body weight and standard length of 5.08±1.28 mg and 9.20±1.56 mm, (n = 5), respectively were used in the study. Weight and length measurements were taken 5, 10, 15, 20 and 24 days of culture. Length measurements were taken by the use of a calibrated transparent ruler (mm) and weight measurement were taken by a very sensitive electronic meter balance.
Monitoring of Water Quality Parameters
The tanks were monitored daily for fry presence, dissolved oxygen, temperature, pH and ammonia using methods described by APHA (1985). Total ammonia nitrogen were determined at 5 day interval by the Nessler method with a spectrophotometer un-ionized ammonia levels were determined with equations given in Emerson et al. (1975).
Indices of Growth and Mortality Rates Specific Growth Rate was calculated using the formula;
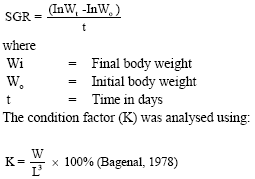 |
Survival rates, mean final standard length and mean body weight were compared using single classification ANOVA followed by Duncan’s Multiple Range Tests (Duncan, 1955).
RESULTS
Results of the water quality parameters in the test tanks are presented in Table 1. Water quality differed only slightly among the various treatments. Water temperature, pH, TAN and ionized ammonia did not vary among the treatments (p<0.05). The treatment with enriched rotifer recorded slightly lower dissolved oxygen levels during the experimental period.
The percentage survival, growth and specific growth rate (SGR %) of the African Catfish larvae fed the rotifer, B. calyciflorus in comparison to a mixed Zooplankton diet is given in Table 2. Percentage survival was slightly high in all treatments (39.0-68.0%). However, higher percentages of 62.0 and 68.0% were reported for larvae raised on the freshwater rotifer and enriched rotifer, respectively. There was however no significant difference in all the treatments. It was observed that fry mortalities decreased drastically after 15 days of rearing period.
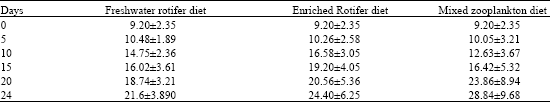
Mean weight increase did not show much difference as it ranged from (93.47-174.72 mg); however fry fed with a mixed Zooplankton diet had higher growth rates than those raised with rotifers alone. Analysis of Variance (ANOVA) revealed no significant difference in the larvae raised with the different live food (p>0.05). Table 3 and 4 shows the mean weight and length of the African catfish C. anguillaris larvae through 24 day rearing period, respectively. Weight and length increment in the age specific growth of the cultured larvae were linear in all the treatments. Larva attained a weight of range between 48.0 and 78.46 mg and length of 16.02 and 19.20 mm after 15 days of rearing period. Although larvae fed mixed zooplankton diet had better growth in terms of length and weight increase but survival was generally low as compared to larvae raised on rotifers diets. Table 3 and 4 also showed that growth increase for the larvae within the first 10 days were better in the rotifer fed larvae (36.0-38.62 mg) than for the mixed zooplankton diet fed larvae (12.63 mg). However as the culture period progressed to 20 days, the fry fed mixed zooplankton diet increased more in both their weight and length.
| Table 1: | Some physical and chemical parameters from tanks stocked with African catfish fry and fed with the Freshwater rotifer, B. calyciflorus, enriched rotifer and mixed zooplankton diets |
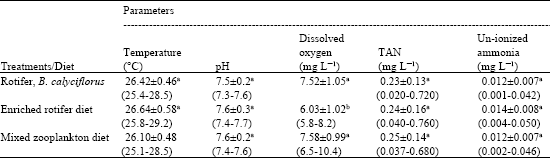 | |
| Values are Mean±SD and Range (in Parentheses). Vakues in a column followed by the same letter are not significantly different at p<0.05 using Duncan Multiple range test | |
| Table 2: | Percentage survival, growth and specific growth rate (SGR%) of the African catfish Clarias anguillaris larvae fed the freshwater rotifer, B. calyciflorus in comparison to mixed zooplankton diet |
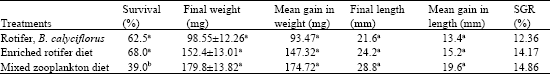 | |
| Initial body weight = 5.08±1.28 mg, Initial length = 9.20±2.56 mm. Values in a column followed by the same letter are not significantly different at p<0.05 | |
| Table 3: | Mean+SE of total weight increase of Clarias anguillaris larvae through 24 days rearing period with different diets |
 | |
| Table 4: | Mean+SE of total length increase of Clarias anguillaris larvae through 24 days rearing period with different diets |
 | |
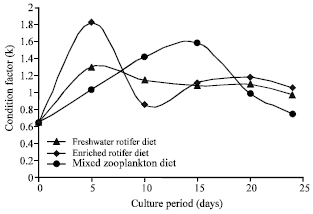 | |
| Fig. 2: | Fluctuation in the mean condition factor (K) of Clarias anguillaris larvae fed rotifer in comparison to a mixed zooplankton diet through 24 days culture period |
Specific Growth Rate (SGR) of C. anguillaris fry fed mixed Zooplankton diet was significantly higher than rotifer fed fry (x¯ = 1.07) over a 24 day rearing period.
The values of the condition factor (K) indicated that it was fairly constant in all treatments (Fig. 2).
DISCUSSION
From the results obtained in this investigation, the freshwater rotifer B. calyciflorus can be isolated effectively using organophosphorus acid ester (diazinon) at the rate of 1.2 mg L-1. This concentration which was arrived after series of toxicity experiments as reported in another investigation by Agbon et al. (2002) in the same laboratory. Several other chemicals such as trichlorfon, fenthion and diflbenzuron in the past have been reported and found effective for initiating pure cultures of rotifer (Burtle and Morrison, 1987; Ludwig, 1993). Stable continuous culture of the freshwater can be maintained by employing the ‘modified feed back mechanism’ adopted in this investigation.
The water quality parameters in the various tanks fluctuated slightly. These values are all within acceptable range for larvae rearing (Ottera, 1993; Ludwig, 1994; Lubzens et al., 2001; Arimoro, 2005). Dissolved oxygen levels however dropped in the culture tank with enriched rotifer and larvae. Probably the oil must have prevented the dissolution of oxygen into the water.
Culturing African catfish fry to fingerlings in tanks would allow monitoring of the survival, growth and condition and enable the culturist to maintain optimal temperature and other growing conditions. For commercial fingerling producers it would eliminate the need and stress involved in harvesting fingerlings from ponds and transporting them to holding tanks before they are packed for supply.
The percentage survival of larvae raised on the freshwater rotifer, B. calyciflorus was relatively high as compared with earlier studies. Ludwig (1993) obtained a 21.2% survival rate of fry of sunshine bass offered the freshwater rotifer, B. calyciflorus. This result is an indication that the freshwater rotifer, B. calyciflorus can be successfully used as a starter food for the African catfish larvae. Lim and Wong (1997) also subscribe to this. They opined that the freshwater rotifer, B. calyciflorus was useful during the first two weeks of exogenous feeding in the discus larvae, an ornamental fish species. Similarly, Shiri et al. (2003) obtained a high survival rate (69.2%) of the burbot larvae, Lota lota raised on B. calyciflorus as starter food.
Survival was lower in the fry fed a mixed zooplankton diet of rotifers, copepods and cladocerans (39%), although these larvae had better growth rate in terms of length and weight increase. Many factors must have probably led to the high mortality of these fry which included; that some calanoid groups which are predatory species must have harmed the young larvae (Ludwig and Lochmann, 2000). Also there is the tendency of the larvae feeding on larger optimal food particles to exhibit fast individual growth but low group survival (Ajah, 1998). The sudden outstanding increase in growth of a few fish individual of the same parental origin over others leading to excessive cannibalism on the smaller ones and greater food consumption during competition. The bigger fry were seen occasionally catching the smaller ones. This is not unexpected as most fish larvae are carnivorous. A high percentage of unaccounted for mortalities in the respective mixed zooplankton fed larvae is an indication that many larvae must have starved early because they cannot eat the large prey items and the few survivors that grew, then had the entire experimental ration to eat themselves.
The larva of the African catfish is small at hatching ≤4.00 mg and 7 mm in weight and length, respectively (Jeje, 1992) and strives better on very small zooplankton as the rotifer used in this investigation. Rotifer sizes ranges from <0.1 to 1 mm (Wallace and Snell, 1991). Copepod nauplii are slightly larger than rotifers with adult copepods 0.5 to 2 mm (Williamson, 1991) and adult cladocerans are 0.2 to 18 mm long (Dodson and Frey, 1991). Thus by virtue of their small size, rotifers are usually the first live food for the small fish larvae (Lubzens et al., 2001), also their relative immobility makes it easier for it to be found and captured as food with lower energetic cost (Ludwig and Lochmann, 2000).
Mean weight and length gain of various treatments did not show much difference however it was observed that in the first 10 days of larval culture, larvae fed rotifers had better growth. This is a confirmation that at the early days of life of the fry, rotifers were utilized more for growth than the mixed zooplankton diet. There was a shift from the 15 day of larval culture to day 24; larvae reared on mixed zooplankton had better growth. Probably, the best period for utilizing mixed zooplankton diet is from the 10th day of culture when the fry would have developed larger mouth size and swim faster to obtain the zooplankton (Ajah, 1998).
Filling and fertilizing a series of ponds at times estimated to provide an abundance of rotifers and subsequently copepod nauplii and cladocerans would allow the fish culturist to have a supply of appropriately sized live food available at the different stages of development of fry development.
The best growth and survival was obtained in larvae fed enriched rotifer diet. According to Lubzens et al. (1989), the rapid loss of organic material from rotifers which are deprived of food is generally perceived as one of the main factors causing poor growth and high mortalities in fish larvicultures. This is probably the reson for the use of the green water by most larval culturist, which maintains the rotifers in a healthy nutritious state. Other researchers supported the fact that enriched rotifers fed to larvae enhanced their growth and survival rates (Craig et al., 1994; Rimmer et al., 1994; Castell et al., 2003). Lubzens et al. (1989) reported that in order to ensure adequate amounts of essential lipids, rotifers must be enriched with either an appropriate alga or emulsified freshoils. In addition, Cruz -Hernandez (1999) reported best larval growth and survival by enrichintg rotifers with powersh-fish oil, containing a highly unsaturated fatty acid (HUFA) 22; 6n-3 in the red porgy (Pagrus pagrus) larvae. This is consistent with the findings of the present investigation. It is thus recommended that rotifers should first be enriched in a suitable medium before feeding them to fish larvae.
CONCLUSION
The freshwater rotifer, B. calyciflorus appears to be ideal live food for the first few days’ culture of the African catfish larva because of its numerous characteristics; small size, slow morbidity and easy catchability by the larvae. It is also important to enrich this rotifer for all round best performance in the larva. The culture of the freshwater rotifer, B. calyciflorus can be maintained continuously in a ‘feed back’ culture system adopted in this investigation. After about 10 days of larval culture it is desirable to use mixed zooplankton diet which contain some larger sized organisms that can sustain better growth rates in the fry.
Freshwater larviculturist will avail themselves with the findings of this research in order to improve larval performance, increase yield and facilitate breeding of new fish species. This will ensure an overall satisfactory performance in hatchery operations.
ACKNOWLEDGMENTS
The author is grateful to the Rock water fish farm, Jos for providing the larvae used in this research. Mr. Andrew Agbon is greatly appreciated for helping to establish the safe concentration for obtaining pure cultures of the rotifer used in this research. Professor P.C. Ofojekwu directed this research and the department of Zoology, University of Jos, Jos-Nigeria for providing facilities used for this research.
REFERENCES
- Agbon, A.O., P.C. Ofojekwu, I.S. Ezenwaka and W.O. Alegbeleye, 2002. Acute toxicity of diazinon on rotifer, cyclops, mosquito larvae and fish. J. Applied Sci. Environ. Manage., 6: 18-21.
Direct Link - Arimoro, F.O., 2006. Culture of the freshwatewr rotifer, Brachionus calyciflorus and its application in fish larviculture technology. Afr. J. Biotechnol., 5: 536-541.
Direct Link - Castell, J., T. Blair, S. Neil, K. Howes and S. Mercer et al., 2003. The effect of different HUFA enrichment emulsions on the nutritional value of rotifers (B. plicatilis) fed to larvae haddock (Melanogrammus aeglefinus). Aquacult. Int., 11: 109-117.
Direct Link - Cruz-Hernandez, C.M., M. Salhi, M. Bessonart, M.S. Izquiero, M.M. Gonzalez and H. Fernandez-Palacious, 1999. Rearing techniques for the red porgy (Pagrus pagrus) during larval development. Aquaculture, 179: 489-497.
Direct Link - Lim, L.C. and C.C. Wong, 1997. Use of the rotifer, Brachionus calyciflorus Pallas, in freshwater ornamental fish larviculture. Hydrobiologia, 358: 269-273.
Direct Link - Lubzens, E., O. Zmora and Y. Barr, 2001. Biotechnology and aquaculture of rotifers. Hydrobiologia, 446-447: 337-353.
Direct Link - Ludwig, G.M., 1993. Effects of trichlorfon, fenthion and diflubenzuron on the zooplankton community and on the production of the reciprocal-cross hybrid stripped bass fry in culture ponds. Aquaculture, 110: 301-319.
Direct Link - Ludwig, G.M., 1994. Tank culture of sunshine bass Morone chrysops H M. saxatilis fry with freshwater rotifer B. calyciflorus and salmon starter meal as first food sources. J. World Aquacult. Soc., 25: 337-341.
Direct Link - Ottera, H., 1993. Feeding, growth and survival of Atlantic cod (Gadus morhua) Larval reared in replicate plastic enclosure. Can. J. Fish. Aquat. Sci., 50: 913-924.
Direct Link - Rimmer, M.A., A.W. Reed, M.S. Levith and A.T. Lisle, 1994. Effect of Nutritional enhancement of live food organisms on growth and survival of Barramundi (Lates calcarifer) Larvae. Aquaculture, 25: 143-156.
Direct Link - Wang, C., S. Xie, K. Zheng, X. Zhu, W. Lei and Y. Yang, 2005. Effects of live food and formulated diets on survival, growth and protein content of first-feeding larvae of Plelteobagrus fulvidraco. J. Applied Ichthyol., 21: 210-214.
CrossRefDirect Link








