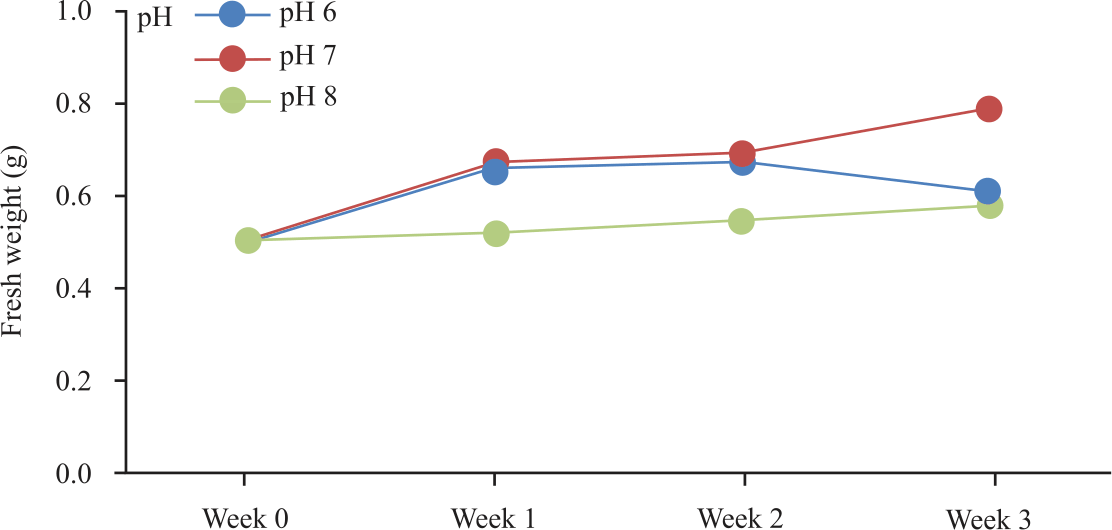
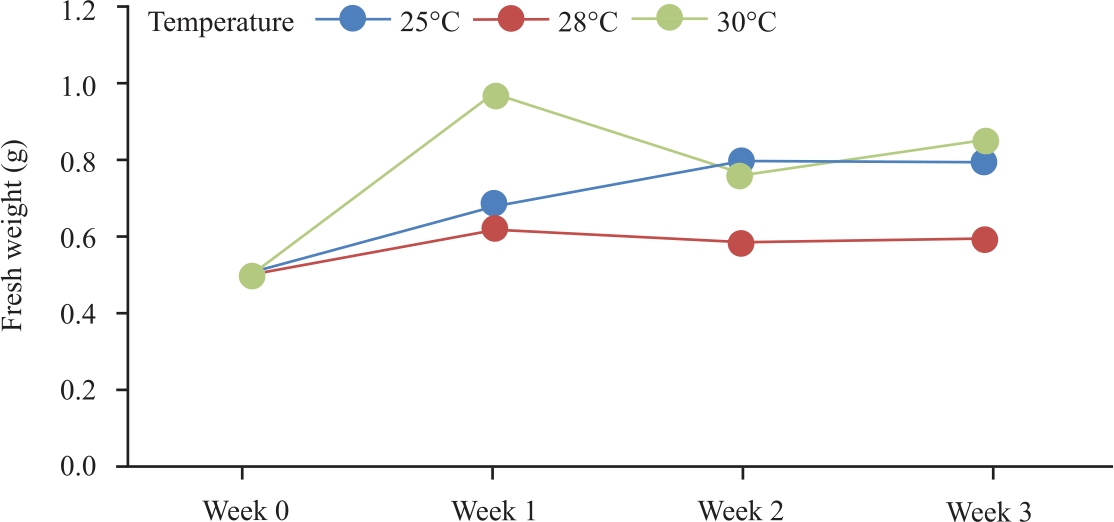
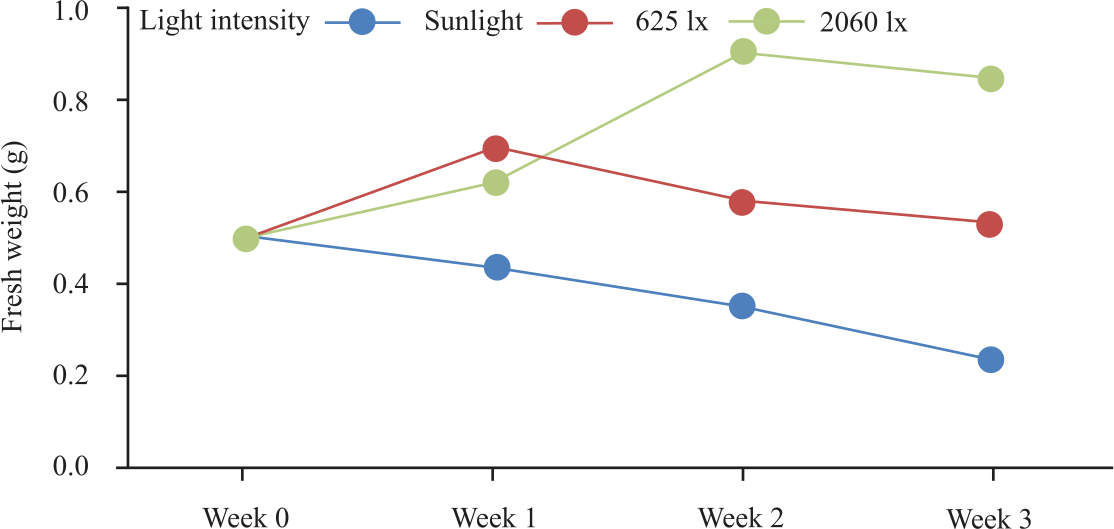
Research Article
Impact of Growth Conditions on Indigenous Algal Biomass Production
Department of Zoology and Environmental Sciences, Punjabi University Patiala, Punjab 147002, India
LiveDNA: 91.25224
Anita Rajor
School of Energy and Environment, Thapar University Patiala, Punjab 147004, India
Amritpal Singh Kaleka
Department of Zoology and Environmental Sciences, Punjabi University Patiala, Punjab 147002, India
LiveDNA: 91.2806