Research Article
Peat Water Purification by Hybrid of Slow Sand Filtration and Coagulant Treatment
Department of Physics, Math and Science, University of Riau, Jalan Binawidya, Pekanbaru, Indonesia
LiveDNA: 62.16600
Peat lands provide many local and global environmental services and benefits all over the world. Urban water consumption is supplied through surface and underground channels. However, communities tend to opt for underground sources owing to the contamination and pollution of surface water. In Riau, especially in urban city, extraction of underground water is preferred to surface sources1,2, because of the numerous lowlands which is covered by swamp or peat. The river contained more organic and acidic compounds3-6. Since time immemorial, excessive exploitation of underground water has given rise to numerous environmental problems such as decreasing land level and sea water intrusion7-9.
Peat water is commonly found in 3 mainland in Indonesia such as in Papua, Sumatera and Borneo Island with an approximate percentage of 3.5%10. Peat region has a lot of ground and surface water sources11. But, it contains a tremendous amount of dissolved organic carbon (DOC) and humid acid, thereby, turning the color dark-brown with lower pH making it not suitable for consumption12-14.
Slow sand technique is a proper cost efficient method that has long been utilized to improve peat water quality in cities. This method is more efficient than rapid sand filter because it consist of fined sand materials and gravels15. Many modifications were conducted by engineers to meet with corresponding water conditions. For example, when water is polluted by iron, adding a coagulant agent is necessary16. However, previous researchers have succeeded in reducing the concentration of iron in polluted water using alum17.
In the most region of Riau province, including Tambang village, the use of water surface is avoid because dominated by peat water, a non-consumable water as we mentioned before. So, the citizen residents used underground water by drill-well which affects to groundwater source for environment in the future. For maintaining the balance of surface and groundwater, surface water source should be a primary source of water supply for the citizen consumptions. So, in the peatland region, there is necessity to purify the peat water firstly before use by the communities, at least for small scale use. This research proposes a hybrid technique of coagulant agent and modified-slow sand filtration for purifying peat water in small size. We examines the water quality of the before and after treatment by analyzes their physical and chemical parameter, that is characterized by improvement in electrical conductivity, pH, heavy metal content, total dissolved solid (TDS) compare to the international and national standard of water quality.
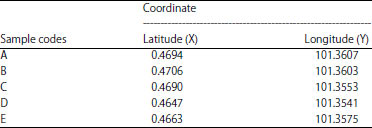
Sampling collection: In this study, the samples are collected from peat water channels and streams in Rimba Panjang village, Kampar Regency, Indonesia from 29 February-5 March, 2018. The sampling coordinates are shown in Table 1.
Treatment processes: In this study, coagulant treatment is combined with slow sand filter. Coagulant treatment is a sample technique used in processing bad water commonly contaminated by pollutant for better quality. Alum is used and dissolved in the tank filled with swamp.
The filter is carried out in PVC pipes of 1.7 m which are filled with several filtering materials consisting of white quartz sand (20 cm), foam filter (15 cm), bio ring stone (5 cm), zeolite (10 cm), charcoal (15 cm), gravel and big stone as illustrated in Fig. 1. Each layer is bordered by strainer sheet of 1 cm, with the sand and foam playing the main role in the filtration process. Zeolite is used as an ion exchange medium in the water purification process. Gravel stones are generally smooth textured and used as last filtration with its size between 2-75 mm and the shape not homogeneous. This big stone (diameter of 2-6 cm) can absorb contaminated water, while charcoal and bio ring stone absorbs bacteria. In the initial step, 8 L of peat water from each sample were loaded into the tank. Then, 3 g of alum are added and stirred for 2-3 min until it is properly dissolved. Next, the air pressure is loaded by pumping manually for about 2 min.
The water was left for 2 h, so that the sediments are settled at the bottom of the tank. After that, the tap was opened and water flowed into slow sand pipe and drop from outlet side into the container as shown in Fig. 2. To measure conductivity, pH and TDS using conventional method, peat groundwater samples must be taken to the laboratory and measured using a conductivity meter, pH meter and TDS meter, respectively.
Physical parameters: The physical parameters measured to determine peat water quality are TDS, electrical conductivity and pH taken from peat water channel and stream.
| Table 1: | Geographical coordinate of sample location |
 | |
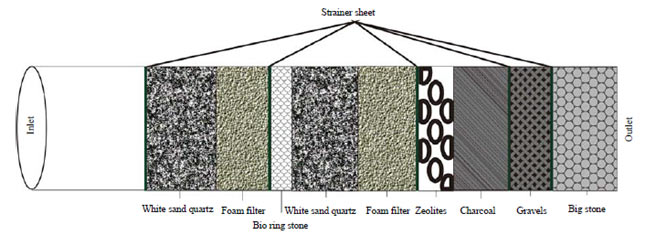 | |
| Fig. 1: | Material arrangement of slow sand filter |
Source: Modification from Logsdon et al.15 | |
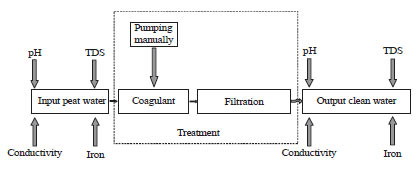 | |
| Fig. 2: | Equipment settings peat water purification |
Source: Modification from Logsdon et al.15 | |
| Table 2: | Standard of consumption water |
 | |
*At 20°C, EC: Electrical conductivity, TDS: Total dissolved solid | |
Quality test data was analyzed by comparing the measured value to the standard accepted by WHO18, European Nation19 and Indonesia national standard20 as given in Table 2.
Electrical conductivity is related to total. This is commonly expressed by linear equation21-23:
| (1) |
where, k is a constant for a particular type of water. For many cases, electrical conductivity-TDS relation are not linear with range value of k is 0.55-0.85. The pH value can influence speciation of chemical compounds and toxicity of trace elements found in water24:
| (2) |
where, pH is the degree of acidity.
Standard deviation technique is used to analyze data from measuring the physical parameters. It can be written as El-Taweel and Ali25:
| (3) |
where, S is standard deviation, xi is physical parameter value, is mean value of the parameters and n is number of physical parameter data.
To find out the optimum discharge, the pipe is set with several angles. As summarized in Table 3, the maximum output rate is obtained by 30° slope where the turbidity is still 0 NTU. We found that over 30°, the turbidity of outlet water is >0 NTU.
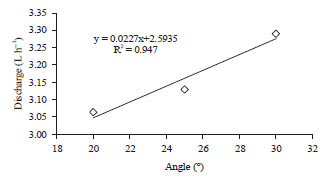 | |
| Fig. 3: | Empirical relationship the tube slope to water discharge with zero turbidity |
 | |
| Fig. 4: | Peat water sample before treatment (brownish) and after treatment with coagulants and slow sand filters (limpid) |
Furthermore, the filtration of all samples is done by same pipe slope, i.e., 30°. The empirical relation of discharge rate and angle of pipe slope is shown by Fig. 3. The relation may be used for predicting discharge rate for slope pipe below 20° where the turbidity remains 0 NTU.
The measured physical-chemical parameters of peat water before and after treatment are given in Table 4 and 5. All the samples are examined for the same volume. All the samples are relatively homogeneous indicating by small deviation values. Furthermore, the concentration of heavy metal, represented by iron (Fe) ion, is measured both for before and after treatment as provided in Table 6. The significant improvement is found in conductivity and pH level. Before treatment, the lower pH of peat water (~3.62) is increased to be 6.79, averagely. Other parameters such as TDS and Fe content is only slightly decreased but still sufficient to the standard18-20.
| Table 3: | Effect of filtration tube slope on discharge with color indicator after filtration and coagulant |
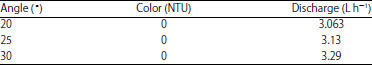 | |
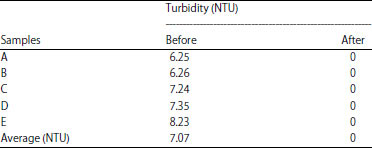
The main issue of peat water is their visual appearance, darker color than freshwater (see 2 left bottles in Fig. 4). So, the water is easily estimated as a non-consumable water source despite the fact that most of Riau province is covered by swamp peatland and of course, peat water source10,11. Fortunately, our filtration technique is successfully worked for eliminating dark-brown (sometimes reddish to yellow) of peat water as seen in Fig. 4 (2 right bottles). More limpid color is obtained after filtration. This claim is also supported by 0 NTU of turbidity of the treated water as given in Table 7.
Based on Fig. 3 it can be seen that the relationship between the slope of the filtration tube and the discharge of the treated water is very significant, with a correlation coefficient of 0.947, this means that there is a very strong relationship between the 2 variables. By using the empirical equation obtained, it can be calculated that the maximum discharge of treated water is 6.68 L h–1.
Table 4 and 5 is clearly proven that the samples have a lower mineral ion indicating by low electrical conductivity, while the pH is lower. The acidic compounds, such as carboxylic and phenolic acid,21 is created more hydronium ion (H+) and lower pH. These chemical and physical properties are the main reason why it was not recommended for drinking. Although some communities in these affected areas may tolerate this condition, normally people will not consume it if there are freshwater resources available.
After treatment, the physical appearance of the sample is better as shown in Table 4 and Fig. 4. The coagulant works properly to agglomerate most pollutant matter. The working mechanism of this coagulation is completely described by El-Dardeer et al.16. The electrical conductivity of the samples extremely rises to about 517% because the contribution of alum and mineral components is slow sand filter as indicated by TDS increment. However, the conductivity of both before and after treatment remains acceptable for drinking base on European standard19 as depicted in Table 4 and 5. The standard considered by WHO and National Indonesia for electrical conductivity of consuming water may vary for different geological regions19. Electrical conductivity is widely used to test water quality assessment.
| Table 4: | Physical-chemical parameters of peat water before treatment |
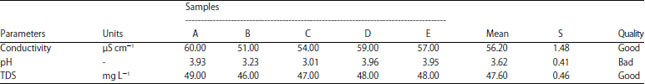 | |
S: Standard deviation over all samples | |
| Table 5: | Physical-chemical parameters of peat water after treatment |
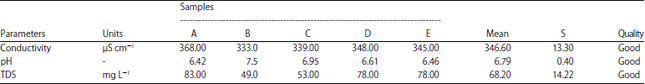 | |
S: Standard deviation over all samples | |
| Table 6: | Concentration of iron before and after treatment |
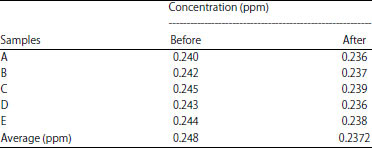 | |
| Table 7: | Turbidity of the samples before and after treatment |
 | |
Higher conductivity value indicates that the water is polluted by heavy metal5. In this study, the highest electrical conductivity is found in sample A because it is located close to the household waste channel.
Reddish or darker-brown color is also typical for peat water21,23 as depicted on the left side of Fig. 4. After filtration, the turbidity of the peat water is to be 0 and limpid appearance is obtained (right side of Fig. 4). This result is really improved the peat water before treatment which has average turbidity of 7.07 NTU and better improvement than slow sand filtration which developed by El-Taweel and Ali25, they obtained water turbidity after filtration is 0.5.
It is well known that tropical peatland contains more humid acid and organic contents23 and it is supported by the pH of all samples before treatment averaging 3.62. By applying the treatment, the acidic level of each sample can be increased significantly to 6.79. This is carried out using the neutralization process of hydronium ions produced by slow sand filters, especially zeolites. Moreover, iron concentration after treatment is narrow different of 2.3% than before treatment as shown in Table 5. This value is lower than 3 mg L–1, so the turbidity may not be affected by iron concentration. There are no concentration standards for iron but the higher concentration may contain iron.
Several physical and chemical properties of peat water are improved using hybrid coagulant slow sand filter which opens a development of clean supply in these regions, especially in Riau Province which is dominated by drilled-ground sources. Excessive exploitation of underground water can contribute to decreasing land level from the sea in the future16,26. In this study we have demonstrated a novel of peat water purification using dual treatment-alum and slow sand filter, which proved to improve its quality. Unfortunately, hazardous chemical compounds and microbial tests were not conducted by the researchers. This will be accomplished in future work together with scaling up the capacity of peat water purification.
A method for treating peat water using a slow sand filter combined with a coagulant agent has been carried out. The results show that physical and chemical parameters, such as conductivity, pH and TDS values, can be improved so that they are suitable for consumption. The visual appearance of the treated water is more limpid with zero turbidity. This study opens up the water supply opportunities for people in the peatland area to develop sustainable surface management.
This article describes a new method for producing clean water obtained from peat water raw materials. The novelty of this method lies in the arrangement of the material as filtration and also the finding of an empirical equation between the angle of the filtration tube to the resulting water discharge. The proposed method provides a fast and inexpensive tool for sustainable management of peat water resources.
The authors would like to thank at the University Of Riau for the assistance in data processing at the physics laboratory of the Earth Department of Physics Math and Science Faculty University of Riau.