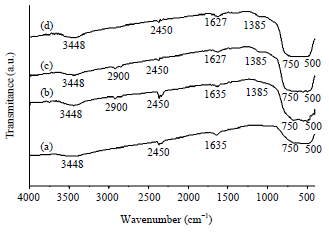
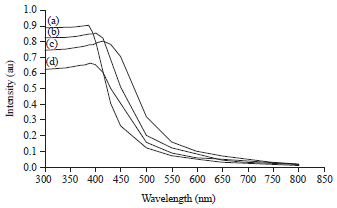
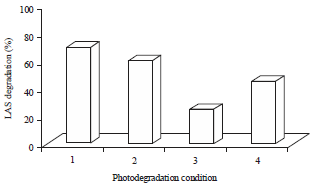
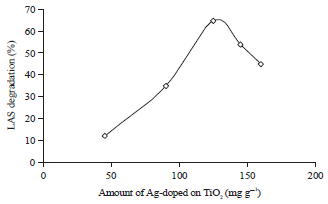
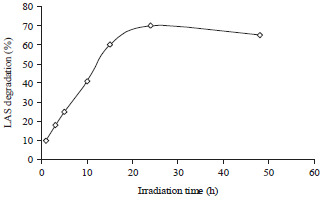
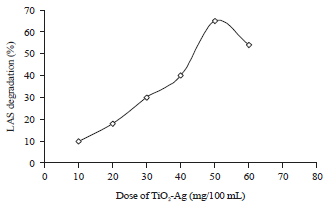
Research Article
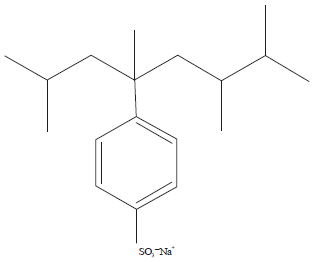
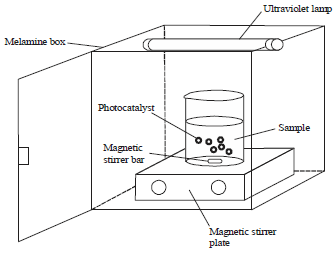
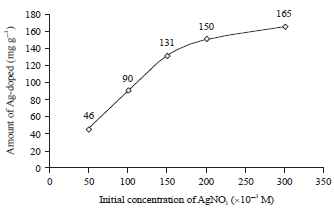
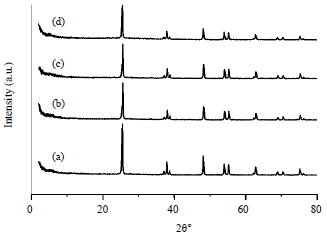
Use of Visible Light for Photo Degradation of Linear Alkyl-benzene Sulfonate in Laundry Wastewater over Ag-doped TiO2
Department of Chemistry, Faculty of Mathematic and Natural Sciences, Universitas Gadjah Mada, Sekip Utara, P.O. Box BLs 21, Yogyakarta, 55284 Indonesia
I. Istiningsih
Department of Chemistry, Faculty of Mathematic and Natural Sciences, Universitas Gadjah Mada, Sekip Utara, P.O. Box BLs 21, Yogyakarta, 55284 Indonesia
A. Suratman
Department of Chemistry, Faculty of Mathematic and Natural Sciences, Universitas Gadjah Mada, Sekip Utara, P.O. Box BLs 21, Yogyakarta, 55284 Indonesia