Research Article
Sustainable Approach for Pharmaceutical Wastewater Treatment and Reuse: Case Study
Water Pollution Research Department, National Research Centre, El-Bohouth St. 33, Dokki, P.O. Box 12622, Giza, Egypt
LiveDNA: 20.17980
I. Abdelfattah
Water Pollution Research Department, National Research Centre, El-Bohouth St. 33, Dokki, P.O. Box 12622, Giza, Egypt
M.E. Abuarab
Department of Agricultural Engineering, Faculty of Agriculture, Cairo University, El-Gammaa Street, P.O. Box 12613, Giza, Egypt
E. Mostafa
Department of Agricultural Engineering, Faculty of Agriculture, Cairo University, El-Gammaa Street, P.O. Box 12613, Giza, Egypt
K.M. Aboelghait
Water Pollution Research Department, National Research Centre, El-Bohouth St. 33, Dokki, P.O. Box 12622, Giza, Egypt
M.H. El-Awady
Water Pollution Research Department, National Research Centre, El-Bohouth St. 33, Dokki, P.O. Box 12622, Giza, Egypt










Hlaing Hlaing Oo Reply
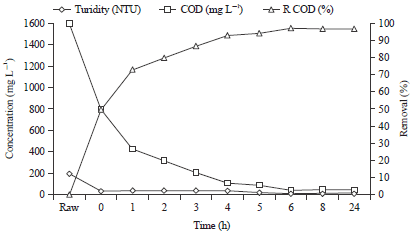
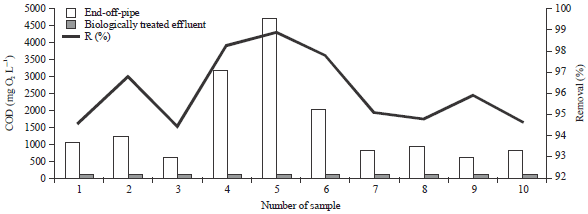
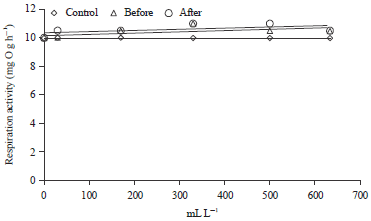
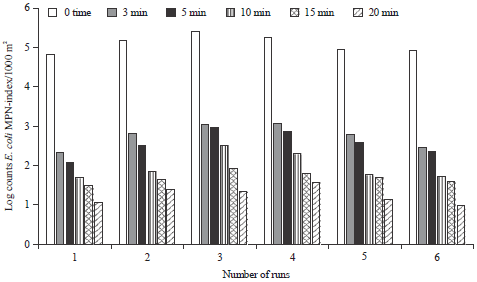
I was interested in pharmaceutical waste water treatment research