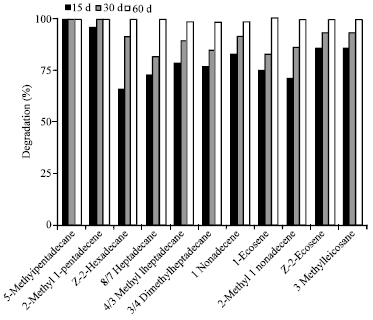
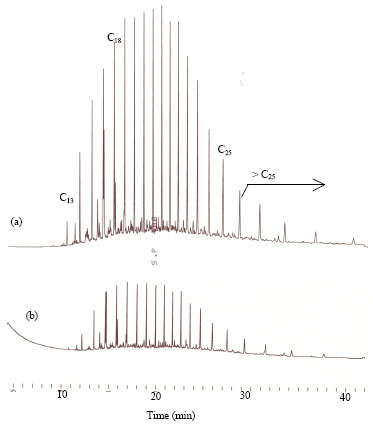
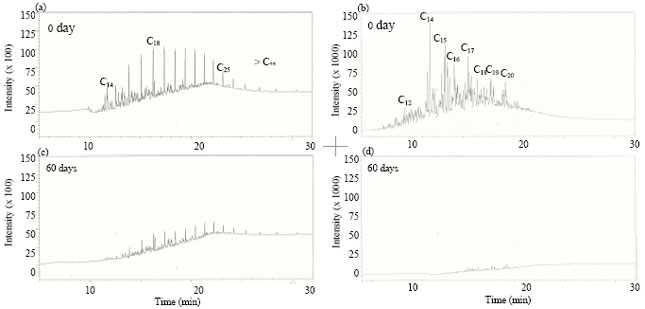
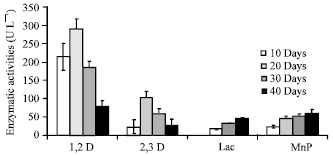
Research Article
Biodegradation of Aliphatic Hydrocarbon in Three Types of Crude Oil by Fusarium sp. F092 under Stress with Artificial Sea Water
United Graduate School of Agricultural Sciences, Ehime University, 3-5-7 Tarumi Matsuyama, Ehime 790-8566 Japan
S. Tachibana
Department of Applied Bioscience, Faculty of Agriculture, Ehime University 3-5-7 Tarumi, Matsuyama, Ehime, 790-8566, Japan