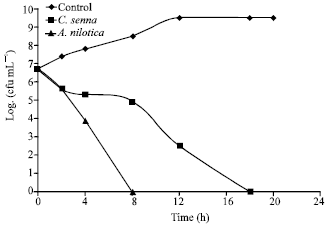
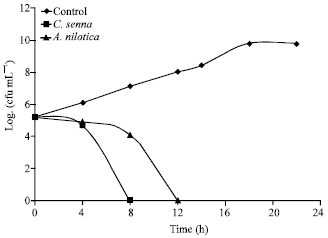
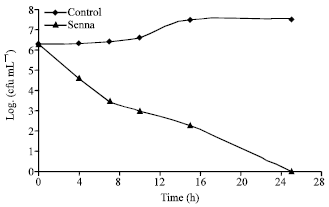
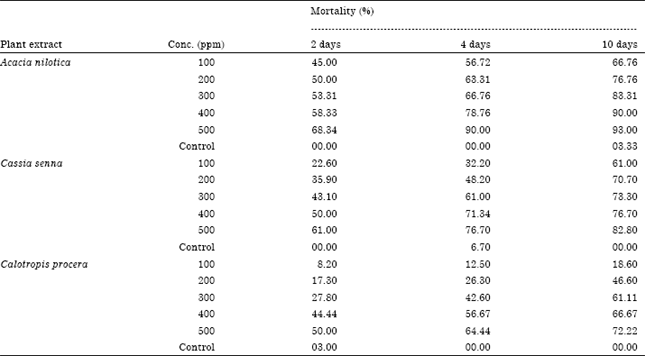
Research Article
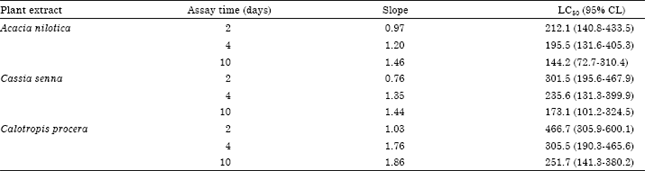
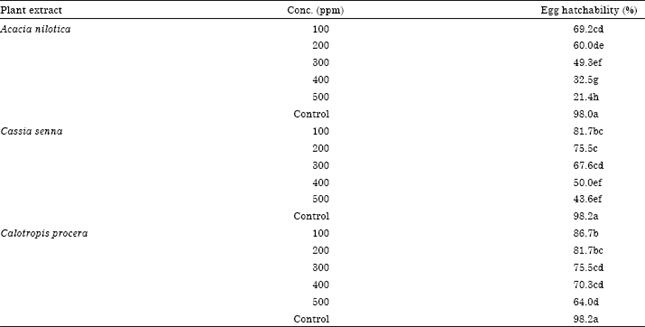
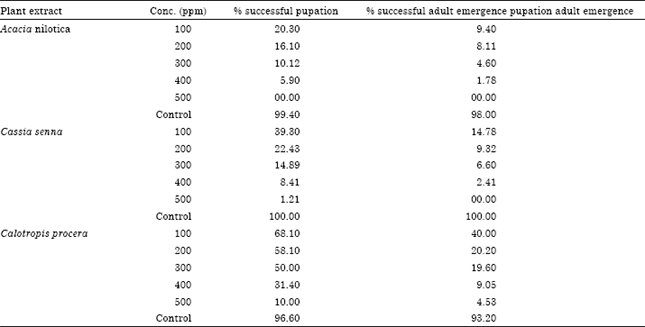
Effect of Three Plants Extracts on Some Bacterial Strains and Culex pipiens L. Stages
Department of Arid Land Agriculture Sciences, Faculty of Meteorology, Environment and Arid Land Agriculture, King Abdul-Aziz University, Saudi Arabia
M.H. Madkour
Department of Environmental Sciences, Faculty of Meteorology, Environment and Arid Land Agriculture, King Abdul-Aziz University, Saudi Arabia
M.Y. Shamy
Department of Environmental Sciences, Faculty of Meteorology, Environment and Arid Land Agriculture, King Abdul-Aziz University, Saudi Arabia