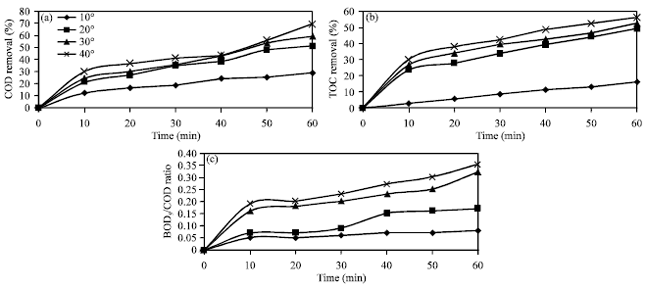
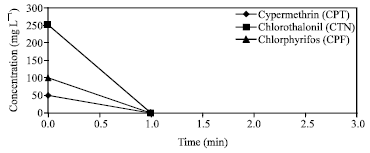
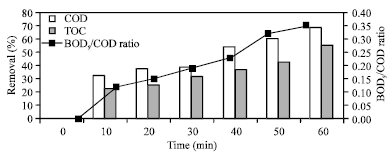
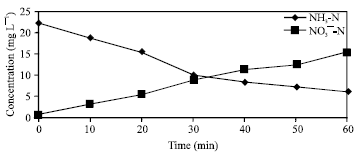
Research Article
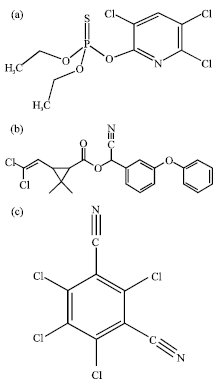
Fenton Treatment of Chlorpyrifos, Cypermethrin and Chlorothalonil Pesticides in Aqueous Solution
Department of Civil Engineering, Universiti Teknologi PETRONAS, Bandar Seri Iskandar, 31750 Tronoh, Perak, Malaysia
Malay Chaudhuri
Department of Civil Engineering, Universiti Teknologi PETRONAS, Bandar Seri Iskandar, 31750 Tronoh, Perak, Malaysia
Shamsul Rahman M. Kutty
Department of Civil Engineering, Universiti Teknologi PETRONAS, Bandar Seri Iskandar, 31750 Tronoh, Perak, Malaysia