Review Article
Extraction of Biodiesel from a Second Generation Energy Crop (Jatropha curcas L.) by Transesterification Process
North South University, Dhaka, Bangladesh
S. A. Sunny
University of Kansas, Kansas, USA
Jatropha curcas L. belongs to the Euphorbiaceous family and receives a lot of attention as a viable source of biodiesel and an alternative to various fossil fuel sources such as petroleum diesel. However, like the latter, it does not emit high amounts of CO2 in the earth’s atmosphere and is considered a renewable energy source. Apart from biodiesel, other energy crops such as sweet sorghum can provide energy in the form of bioethanol (Nahar, 2011). The multipurpose, oil bearing plant originated in Central and South America but currently grow all over the world (Openshaw, 2000). It is a drought-resistant plant and can survive well even when the soil is in water stressed condition due to osmotic adjustment. Osmotic adjustment allows cell to maintain the turgor which is essential for plant growth and various other physiological processes (Nahar and Gretzmacher, 2002). The plant can be propagated easily and established in a wide variety of soil. Different soil types include sandy loam, clay silty soil (Nahar et al., 1993) and soils that have high salinity (Nahar et al., 1989). A common constraint on the widespread acceptance of bioenergy is the space requirement for growth which may lead to future food shortage for the growing global population. Jatropha does not need traditional cultivable land to produce oil-bearing seed which is why it does not contribute to food scarcity or land competition. The plant can reach a height of up to 6 m and has seed yield ranging from 7.5 to 12 tons ha-1 per year. The plant also has a long life of around 50 years, over the period of which it consistently produces oil bearing seeds without any major maintenance.
The oil content ranges from 25-37% in the seeds. The oil contains 21% saturated fatty acids and 79% unsaturated fatty acids. There are some chemical elements in the seeds, like saccharose (or sucrose C12H22O11), raffinose (C18H32O16), stachyose (C24H42O21), glucose (C6H12O6), fructose (C6H12O6), galactose (C6H12O6), protein and oil, largely of oleic- and linoleic-acids, curcasin, arachidic-, linoleic-, myristic-, oleic-, palmitic- and stearic-acids (Perry, 1980).
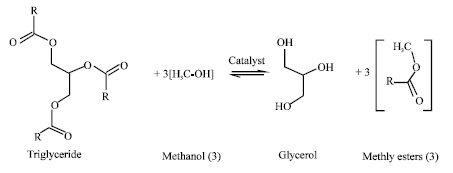
Many world nations have been facing severe energy crisis due to present fossil fuel price and negative environmental impacts. Renewable energy is an alternative option for agriculture based developing countries and developed nations alike to reduce the energy crisis, gain energy security and sequester high amounts of carbon in the atmosphere. The chemical process through which biomass to fuel conversation takes place is called Transesterification which produces fuel grade methyl ester known as biodiesel. In the process, an alcohol is reacted with the triglyceride oils contained in vegetable oils/animal fats or recycled greases, forming fatty acid alkyl esters (biodiesel) and glycerin.
Transesterification: Transesterification converts vegetable oil to biodiesel. In this process vegetable oil is chemically reacted with alcohol in the presence of a catalyst (Ibeto et al., 2011). Cooking oil can also be turned into Biodiesel using this method (Omar, 2009). After the chemical reaction, various components of vegetable oil break down to form new compounds. The triglycerides are converted into alkyl esters which is the chemical name for biodiesel. If methanol is used in the chemical reaction, methyl esters are formed, while if ethanol (C2H5OH) is used, ethyl esters are formed. Both these compounds are biodiesel fuels with different chemical combination. In the chemical reaction, alcohol replaces Glycerin (C3H5 (OH)3). Glycerin that has been separated during the process is released as the byproduct of the chemical reaction. Glycerin will either sink to the bottom of reaction vessel or come to the surface depending on its phase. It can be easily separated by the centrifuges. It can then be used as an energy source for commercial uses (Cerrate et al., 2006). The biodiesel produced by transesterification has much lesser viscosity which makes it capable of replacing petroleum diesel in diesel engines. In the earlier years when process of transesterification was not known the viscosity of vegetable oil was the major hindrance for its use as the fuel for engine. Transesterification process has been able to remove this hindrance. During transesterification process nothing is wasted, as all the products and byproducts can be utilized for different uses. The reaction requires heat and a strong base catalyst, such as sodium hydroxide (NaOH) or more commonly potassium hydroxide (KOH) (Syam et al., 2009). The simplified transesterification reaction is shown as:
It is a simple chemical reaction which neutralizes the “Free Fatty Acids” (FFA) present in any or all fatty substances available from vegetable/animal sources. A chemical exchange takes place between the ‘Alkoxy’ groups of an ester compound by an alcohol. It takes about 10% methyl alcohol by weight of the fatty substance to start the transesterification process.
Plant/animal fats and oils are typically made of triglycerides which are esters of free fatty acids with the Trihydric alcohol, glycerol. In the transesterification process, the alcohol is deprotonated with a base to make it a stronger Nucleophile. Commonly, as can be seen, the reaction has no other inputs than the triglyceride and the alcohol.
Normally, this reaction will proceed either exceedingly slowly or not at all. Heat, as well as an acid or base are used to help the reaction proceed more quickly. It is important to note that the acid or base are not consumed by the transesterification reaction, thus they are not reactants but act as catalysts.
Almost all biodiesel is produced from virgin vegetable oils using the base-catalyzed technique as it is the most economical process for treating virgin vegetable oils, requiring only low temperatures and pressures and producing over 98% conversion yield (provided the starting oil is low in moisture and free fatty acids). However, biodiesel produced from other sources or by other methods may require acid catalysis which is much slower (Ataya et al., 2007). Since it is the predominant method for commercial-scale production, only the base-catalyzed transesterification process will be described below.
Alcohols such as methanol (CH3OH), ethanol (C2H5OH) and even propanol (CH3CH2CH2OH or (CH3)2CHOH), butanol (C4H9OH) and amyl alcohol (C5H11OH) can be used in the transesterification process. Methanol and ethanol are used most frequently, especially methanol because of its low cost and physical and chemical advantages. They can quickly react with triglycerides and sodium hydroxide is easily dissolved in these alcohols. Stoichiometric molar ratio of alcohol to triglycerides required for transesterification reaction is 3:1. In practice, the ratio needs to be higher to drive the equilibrium to a maximum ester yield. The transesterification reaction is base catalyzed. Any strong base capable of deprotonating the alcohol will work (e.g., Sodium Hydroxide (NaOH), Potassium Hydroxide (KOH), Sodium Methoxide (CH3ONa), etc.,). Commonly the base (KOH, NaOH) is dissolved in the alcohol to make a convenient method of dispersing the otherwise solid catalyst into the oil. The ROH needs to be very dry. Any water in the process promotes the saponification reaction, thereby producing salts of fatty acids (soaps) and consuming the base and thus inhibits the transesterification reaction. Once the alcohol mixture is made, it is added to the triglyceride. The reaction that follows replaces the alkyl group on the triglyceride.
An example of the transesterification reaction equation, shown in skeletal formulas where the Triglyceride Oil with Methanol and a catalyst yields Methyl Esters (Biodiesel) with Glycerol as a byproduct.
 |
Jatropha oil is blended with alcohol and catalyst mixture which is kept at reaction temperature for specific duration under agitation and sent to the settling tank. The ester is collected and washed to get pure biodiesel. The pilot plant consists of reaction vessel with heating and agitating device, catalyst mixing vessel, settling tanks and washing tank. The Capacity of pilot biodiesel plant is 250 liters/day (Department of Bio-Energy, 2010). Figure 1 shows a general transesterification process flow where Methanol (CH3OH) and a catalyst (NaOH) are introduced to filter plant oil which yields methyl ester (Biodiesel) after purification and Glycerin as a byproduct. Methanol can be recovered and applied back at the initial phase of separation during transesterification.
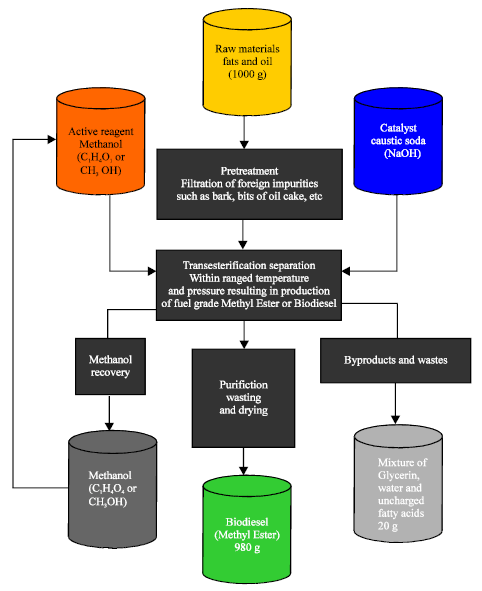 | |
| Fig. 1: | Process flowchart for biodiesel production from Jatropha seeds and by products |
Advantages of biodiesel from Jatropha: Biodiesel is non edible and biodegradable. It reduces the emission of harmful products from diesel engine (80% less CO2 and 100% SO2). It has a high Cetane number (measure of fuel ignition quality) than diesel. The high Cetane number of biodiesel contributes to easy cold starting and low idle noise.
It increases the life of diesel engine as it is more lubricating Biodiesel replaces the exhaustic order of engine with a more pleasant smell. Not only that, it has many important uses like oil for rural energy supply, cake for fish, animal feed, organic fertilizer, bio-pesticides, medicine, soap etc. More importantly, 1000 g of Jatropha seeds can produce 980 g of pure biodiesel. However, most optimized practical processes yield around 94% biodiesel (Kabbashi et al., 2009). Figure 2 shows a sample flowchart that shows the initial stage of oil extraction from the seeds which result in crude biodiesel from a transesterification plant which is then washed to produce pure Biodiesel which can be used in diesel engines as a fuel source. The oil can be directly burnt in lamps and stoves (Fig. 2). The residue from the seeds during and after the oil extraction process can be used to make oil cake and manure and even be used as fodder or cattle feed after removing toxins. Similarly, during transesterification, crude glycerol is formed which can be used to make soap and candles.
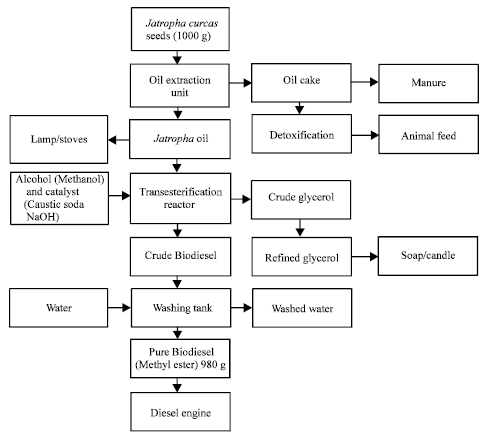 | |
| Fig. 2: | Process flowchart for biodiesel production from Jatropha seeds and by Products |
| Table 1: | Comparison of fossil diesel and Bio-diesel (Heller, 1996) |
 | |
Alcohol and a catalyst is required for successful reaction at the transesterification stage, as discussed earlier.
Compared to fossil fuel, Jatropha oil has a comparable density with a lower viscosity but higher flash point and cetane number, as shown in Table 1.
Jatropha biodiesel is an ideal solution to meet current and future energy demands. As an alternative fuel for diesel engines, is becoming increasingly important due to diminishing petroleum reserves and the environmental consequences of exhaust gases from petroleum-fuelled engines. Biodiesel which is made from renewable sources, consists of the simple alkyl esters of fatty acids. As a future prospective fuel, biodiesel has to compete economically with petroleum diesel fuels.
One way of reducing the biodiesel production costs is to use the less expensive feedstock containing fatty acids such as inedible oils. The availability and sustainability of sufficient supplies of less expensive feedstock will be a crucial determinant delivering a competitive biodiesel to the commercials filling stations. Fortunately, inedible vegetable oils, mostly produced by seed-bearing trees and shrubs can provide an alternative. With no competing food uses, this characteristic turns attention to Jatropha curcas which grows in tropical and subtropical climates across the developing world.