Research Article
Metal Hyperaccumulation in Plants: A Review Focusing on Phytoremediation Technology
Centre for Advanced Studies in Botany, School of Life Science, North-Eastern Hill University, Shillong-7930 22, India
In recent years, public concerns relating to ecological threats caused by heavy metal (HM) have led to intensive research of new economical plants based remediation technologies. Conventional methods used for reclamation of contaminated soils, namely chemical, physical and microbiological methods, are costly to install and operate (Danh et al., 2009). The rapid increase in population coupled with fast industrialization growth causes serious environmental problems, including the production and release of considerable amounts of toxic waste materials into environment (Zhuang et al., 2007). Over recent decades, the annual worldwide release of heavy metals reached 22,000 t (metric ton) for cadmium, 939,000 t for copper, 783,000 t for lead and 1,350,000 t for zinc (Singh et al., 2003). Vanadium released from fertilizer effluents usually ranges between 16.58-18.44 mg L-1 (Sarma, 2008). However, the combustion of coal and petroleum-based products is considered as the primary source of vanadium in the environment (Adriano, 1986). According to an estimate, in US approximately 2125 ton of elemental vanadium has escaped in to the environment annkually (Kaplan et al., 1990).
In an ecological research, any metal or metalloid that causes environmental problem which cannot be biologically degraded should be considered as a heavy metal. Heavy metals are natural components of the Earth's crust, but in many ecosystems the concentration of several HMs has reached toxic levels due to consequence of anthropogenic activities. Fifty three elements fall into the category of heavy metal till date and defined as the group of elements whose densities are higher than 5 g cm-3 and recognized as ubiquitous environmental contaminants in industrialized societies (Padmavathiamma and Loretta, 2007). The adaptative responses of plants to HM contaminated environments are efficient processes that include many physiological, molecular, genetic and ecological traits. These traits give certain species the ability to survive or hyperaccumulate the toxic metals. Soil pollution caused by metals is some what different from air or water pollution, because heavy metals persist in soil much longer periods of time than in other compartments of the biosphere (Lasat, 2002). Out of the 92 known elements present on earth crust, only 17 are known to be essential to all living plants for normal growth and 12 are potentially beneficial required only in trace amounts includes silver (Ag), cerium (Ce), chromium(Cr), fluoride (F), iodine(I), lanthanum (La), rubidium (Rb), tin (Sn), serium (Sr), titanium (Ti), vanadium (V) and tungsten (W) ( Pilon-Smits et al., 2009).
Mechanisms of carcinogenicity has been discussed for metals and classified arsenic, antimony, beryllium, cadmium, chromium, cobalt, lead, nickel and vanadium are some metals and metalloids that are carcinogenic to humans or considered to be carcinogenic to humans by International Agency for Research on Cancer (Beyersmann and Hartwig, 2008). Toxic heavy metals cause DNA damage and their carcinogenic effects in animals and humans are probably caused by their mutagenic ability (Knasmuller et al., 1998; Baudouin et al., 2002). Exposure to high levels of these metals has been linked to adverse effects on human health and wildlife.
The hyperaccumulation of heavy metals in some plants has been recorded by many researchers during last few decades (Barman et al., 2000) and this has emphasized the importance of further advanced research in molecular basis of phytoremediation technology. The hyperaccumulation of heavy metals is depends on the plant species, soil condition (pH, organic matter content, cation exchange capacity etc.) and types of heavy metal (Barman et al., 2001; Spinoza-Quinones et al., 2005; Xian and Shokohifard, 1989; Otte et al., 1993). In metal biology, it is experimentally proved that even some metals that are essential for the normal plants growth (such as iron and copper) may become toxic, depending on the oxidation state, complex form, dose and mode of exposure (Beyersmann and Hartwig, 2008).
The removal and recovery of heavy metals from contaminated media are of great importance in terms of protection of the environment as well as abatement of heavy metal toxicity (Kim et al., 2004). Metals and their compounds are accumulating up to harmful levels, unlike organic waste by some living plants. The level of toxic metals (Pb, Cr, Hg etc.) can be reduced from contaminated media by a number of aquatic plants taken up by the roots system and transported to the stems and leaves without showing toxicity syndrome has confirmed by many studies (Rai et al., 1995; Cardwell et al., 2002). This uptake of metals largely depends on the type and chemical speciation of metal and habitat characteristics of plants, i.e., terrestrial, aquatic etc. Hence, plant selection is important for remediation of contaminated site.
Remediation of metal-contaminated soils became a goal for many research laboratories in the world. The use of plants in designing low cost treatment system is still a challenge in environmental managements. Phytoremediation, a new green technology has been inventoried, in which some metal hyperaccumulator plants were utilized to decontaminate soil, water and ambient environment (Prasad, 2003) and it is growing without symptoms of toxicity (Reeves and Brooks,1983; Baker and Brooks,1989; Baker et al., 1991).
Hyperaccumulator plants represent a resource for remediation of metal polluted site, as they are able to extract wide range of metals and to concentrate them in their upper parts with the character of metal tolerance. In some plant species, the concentrations accumulated in aboveground biomass of metals or metalloids are more than one and up to four, orders of magnitude higher than in other adjacent plants (Baker and Brooks, 1989; Reeves and Baker, 2000). This unique extent of accumulation of heavy metals, have been reported in a total of approximately 500 plant species to date (Kramer, 2010). The first field trial on Zn and Cd phytoextraction was conducted by Baker et al. (1991) and now a days, this technology is receiving considerable attention for clean up of soil contaminated with heavy metals (USEPA, 2000). Several comprehensive reviews have been published, summarizing many important aspects of this novel green technology (Padmavathiamma and Loretta, 2007; Kramer, 2010). The present reviews give general guidance of research trends in last few decades, focusing the physiological and molecular basis of phytoremediation. For this review potential publications were searched using Biological Abstracts (BIOSIS), Biological Sciences (CSA), Biosis Previews (BIOSIS), CAB Abstracts (CAB International), Plant Science (CSA) and Web of Science (ISI) data bases; the last round of the search was conducted in June 2010. We used key words that referred to phytoremediation for searching (different combinations of keywords: metals *, plants, hyperaccumulator*, physiology*, Phytochelatins biochemical*). These keywords produced a reasonable output that was then sorted manually; However, the list is not exclusive. In addition, we checked the reference sections of all discovered publications for references to other potentially suitable studies as a cross reference. This review specially focused update information so far available and provides a critical overview of the present state of the art.
Definitions: The term phytoremediation (phyto meaning plant and the Latin suffix remedium meaning to clean or restore) refers to a diverse collection of plant-based technologies that use either naturally occurring, or genetically engineered, plants to clean contaminated environments (Flathman and Lanza, 1998). Phytoremediation is clean, simple, cost effective, non-environmentally disruptive (Wei et al., 2004) green technology and most importantly, its by-products can find a range of other uses (Truong, 1999, 2003).
Phytoremediation is an eco friendly approach for remediation of contaminated soil and water using plants comprised of two components, one by the root colonizing microbes and the other by plants themselves, which accumulates the toxic compounds to further non toxic metabolites. Various compounds viz., organic synthetic compounds, xenobiotics, pesticides, hydrocarbon and heavy metals, are among the contaminants that can be effectively remediated by plants (Suresh and Ravishankar, 2004).
TYPES OF PHYTOREMEDIATION TECHNOLOGY
The four different plant-based technologies of phytoremediation, each having a different mechanism of action for remediating metal-polluted soil, sediment or water:
| • | Phytoextraction: Plants absorb metals from soil through the root system and translocate them to harvestable shoots where they accumulate. Hyperaccumulators mostly used this process to extract metals from the contaminated site. The recoveries of the extracted metals are also possible through harvesting the plants appropriately |
| Table 1: | Cost of different remediation technologies (Glass, 1999) |
 | |
| • | Phytovolatilization: Plants used to extract certain metals from soil and then release them into the atmosphere by volatilization |
| • | Phytostabilization: In this process, the plant roots and microbial interactions can immobilized organic and some inorganic contaminants by binding them to soil particles and as a result reduce migration of contaminants to grown water |
| • | Phytofiltration: Phytofiltration is the use of plants roots (rhizofiltration) or seedlings (blastofiltration) to absorb or adsorb pollutants, mainly metals, from water and aqueous waste Streams (Prasad and Freitas, 2003) |
Limitations: The application of phytoremediation for pollution control has several limitations that require further intensive research on plants and site-specific soil conditions (Danh et al., 2009). It is generally slower than most other treatment viz., chemical, physical and microbiological plants with low biomass yields and reduced root systems do not support efficient phytoremediation and most likely do not prevent the leaching of contaminants into aquatic system. Environmental conditions also determine the efficiency of phytoremediation as the survival and growth of plants are adversely affected by extreme environmental conditions, toxicity and the general conditions of soil in contaminated lands (Danh et al., 2009). In phytoremediation technology, multiple metals contaminated soil and water requires specific metal hyperaccumulator species and therefore requires a wide range of research prior to the applications. Though the phytoremediation is cost effective (Table 1), environment friendly, ability to reclaim heavy metals contaminated site, several limitations also create trouble in implementing the strategy, e.g., metal must be in bio-available form to plants; if metals is tightly bound to the organic portions of the soil, some time it may not be available to plants. Furthermore, if the metals are water soluble, in nature it will pass by the root system without accumulation.
The phytoremediation of mixed heavy metals contaminated soil have conformant with some problem e.g., The cadmium/zinc model hyperaccumulator Thlaspi caerulescens is sensitive toward copper (Cu) toxicity, which is a problem in remediation of Cd/Zn from soils in the presence of Cu by application of this species. In T. caerulescens Cu induced inhibition of photosynthesis followed the sun reaction type of damage, with inhibition of the photosystem II reaction center charge separation and the water-splitting complex (Mijovilovich et al., 2009). Despite some limitations, present day phytoremediation technology are using worldwide and various research laboratories are at present engaged to overcome the limitations.
PLANTS SELECTIONS CRITERIA FOR PHYTOREMEDIATION
Plant species selection is a critical management decision for phytoremediation. Grasses are thought to be excellent candidates, because their fibrous rooting systems can stabilize soil and provide a large surface area for root-soil contact (Kulakow et al., 2000). The selection of plants is possibly the single most important factor for fruitful phytoremediation strategy. The application of indigenous plant species for phytoremediation is often favoured as it requires less management and acclimatizes successfully in native climate conditions and seasonal cycle. However, some exotic plant species may perform better in remediation of specific metals and can be safely used where the possibility of invasive behavior has been eliminated (USEPA, 2000). Some important criteria in selecting plant species for phytoremediation are:
| • | The levels of tolerance with respect to metal known to exist at the site |
| • | The level of adequate accumulation, translocation and uptake potential of metals |
| • | High growth rate and biomass yield |
| • | Tolerance to water logging and extreme drought conditions |
| • | Availability, habitat preference e.g., terrestrial, aquatic, semi-aquatic etc. |
| • | Tolerance to high pH and salinity |
| • | Root characteristic and depth of the root zone |
METAL HYPERACCUMULATORS PLANTS/FAMILIES
Over 500 plant species comprising of 101 families have been reported, including members of the Asteraceae, Brassicaceae, Caryophyllaceae, Cyperaceae, Cunouniaceae, Fabaceae, Flacourtiaceae, Lamiaceae, Poaceae, Violaceae and Euphobiaceae. Metal hyperaccumulation occurs in approximately 0.2% of all angiosperms and is particularly well represented in the Brassicaceae (Kramer, 2010). Recently Environment Canada has released a database Phytorem which compiled a worldwide inventory of more than 750 terrestrial and aquatic plants with potential value for phytoremediation. The study of Co accumulations in 670 species of terrestrial plants showed that leaf Co concentration was in general less than 0.2 ppm, with the exception of Ericales, Euasterids and Asparagales, where 0.3-0.5 ppm of Co was measured (Watanabe et al., 2007). A list of 26 Co hyperaccumulators species belonging to the families of Lamiaceae, Scrophulariaceae, Asteraceae and Fabaceae has been reported (Baker et al., 2000). Zn hyperaccumulation was first discovered in 1865 in Noccaea caerulescens (syn. Thlaspi caerulescens) of Brassicaceae (Reeves and Baker, 2000) and Ni hyperaccumulation, which was first reported in 1948 in Alyssum bertolonii of Brassicaceae (Kramer, 2010). A member of Crassulaceae Sedum alfredii is the only known Cd hyperaccumulator outside the Brassicaceae (Deng et al., 2007). The hyperaccumulation of arsenic (As) has been discovered in only two species of the Brassicaceae (Karimi et al., 2009) apart from a number of pteridophytes (Ma et al., 2001).
METAL HYPERACCUMULATOR MODEL PLANTS
Mixed pollution with heavy metals is characteristic for many spill areas and industrial effluents dumping sites. The danger for the environment and human health from such sites is large and sustainable remediation strategies are urgently needed. Phytoremediation seems to be cheap and environmentally sound option for reclaim the hazardous toxic metals and metalloids. The most important challenge is how to improve the efficiency of phytoremediation by increasing the accumulation of metals in plants, or by improving key plant biological traits that should enhance metal uptake (Wu and Tang, 2009). The heavy metals accumulation on several plants species have been focused viz., Thlaspi sp. (Baker et al., 1994), Brassica sp. (Blaylock et al., 1997; Huang et al., 1997) and Alyssum (Kramer et al., 1996). Heavy metal accumulators is increasing steadily (currently more than 500 plant species) and some are presented in Table 2.
| Table 2: | Examples of some plants and metals they can remediate |
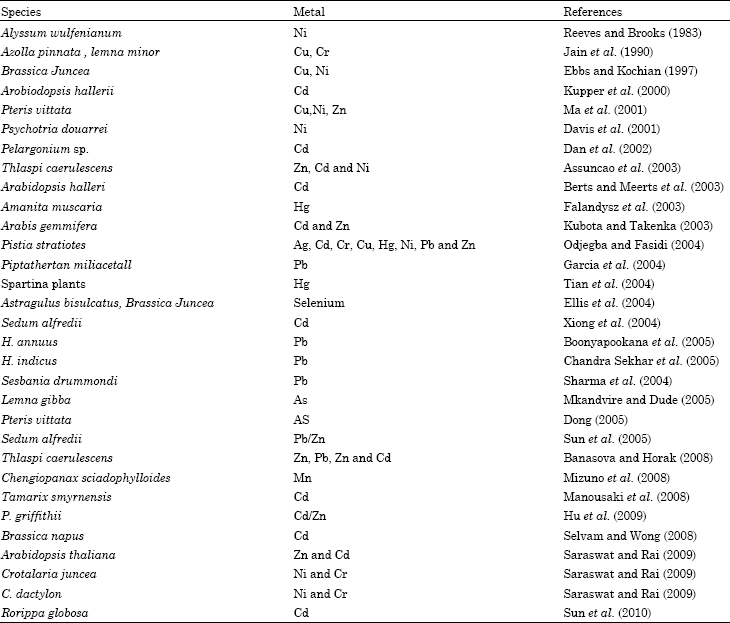 | |
Some species are highly metal specific, have a small biomass, slow growth habit and require careful management for multiplications (Gleba et al., 1999) which are not suitable for commercial applications. Thus identification of novel plant species with high biomass yield coupled with ability to tolerate and accumulate multiple metals has become an important aspect of phytoremediation research. A multiple metal hyperaccumulator scented geranium (Pelargonium sp.) has been recently discovered. This plant has an ability to tolerate and accumulate multiple metals (Cd, Ni and Pb) and maintain normal metabolic processes (Dan et al., 2000; KrishnaRaj et al., 2000). Another potential Pb hyperaccumulator perennial shrub Sesbania drummondii with high biomass yield has been discovered from southern coastal areas of the United State (Sahi et al., 2002) was given as evidence for model plants.
In the last few decades many scientists in different parts of the worlds has worked out the metals bioaccumulation potential of various species and some are presented in Table 3 derived through meta-analysis.
Works on physiological aspects of Vetiveria zizanioides has provided an extensive research foundation which makes this plant a good candidate for a wide range of phytoremediation purposes (Danh et al., 2009).
| Table 3: | Examples of some metal hyperaccumulator and their bioaccumulation potential |
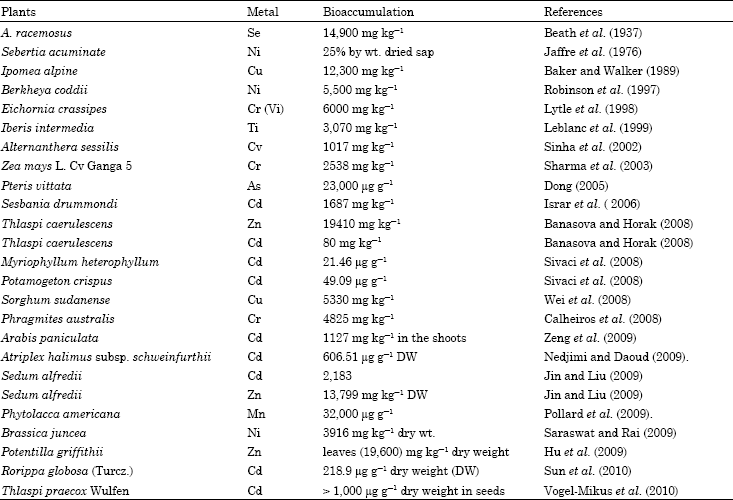 | |
| Table 4: | Concentrations of heavy metals accumulated in Vetiveria zizanioides roots and shoots (Truong, 1999) |
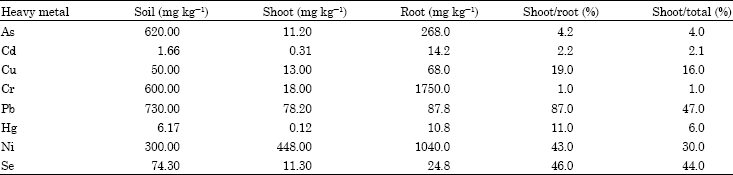 | |
This plant has an ability to accumulate multiple heavy metals in roots and shoots (Table 4) which has been experimentally determined.
The multiple metal accumulation and metal transfer factors from soil (TFS) of three wild macrophyte species viz., Ipomea sp., Eclipta sp. and Marsilea sp. have been studied by Gupta et al. (2008) and recorded that Ipomea sp. shows transfer factor (TFS) for Cd, Cu, Mn and Zn, while Eclipta sp. and Marsilea sp. shows TFS ≥1 for Fe, Cu and Cd. The ratio of metals between soil and plant parts (TFS) is an important criterion for the selection of model plant species for phytoremediation and the ratio >1 means higher accumulation of metals in plant parts than soil (Barman et al., 2000). Among aquatic plants, Elodea densa showed a high accumulation of mercury in leaves, stems and roots from the natural sediment enriched with CH3HgCI (Ochiai, 1987) which make this plant potentials for Hg remediation.
PHYSIOLOGY OF METAL HYPERACCUMULATION
Metals influenced the physiology of plants by promote or inhibit the growth. Some metals that required in high concentration suggest a structural or osmotic role, while effects at low concentration may indicate a role as cofactor for specific enzymes. The summary of the beneficial effects of Al, Co, Na, Se and Si on plants under represented (Fig. 1) adopted from Pilon-Smits et al. (2009).
Many plants developed Al tolerance characteristics via either apoplastic or symplastic detoxification mechanisms (Ma et al., 2001; Pilon-Smits et al., 2009).
In higher plants, Co has been reported to strongly bind to roots and to be mainly absorbed from the soil solution through passive transport. Co is chemically similar with nickel (Ni) and these two elements enter inside the cells through plasma membrane carriers and may be transported by IRT1 (Pilon-Smits et al., 2009). A strong adsorption of Cd on root apoplast might act as a driving force to extract the metal from the soil, compete with the symplastic absorption and contribute to the amount of metals taken up by the hyperaccumulator, at least in its roots (Redjala et al., 2009).
Se hyperaccumulators namely Stanleya sp. and Astragalus sp. can accumulate 1000-15000 ppm (0.1-1.5% Se), even from low external concentrations and this has been enhanced by some specialized transporter. Hyperaccumulators such as Astragalus bisulcatus, Brassica oleracea have a specific selenocysteine methyl transferase, lead to accumulation of Se (Tamaoki et al., 2008)
Toxic metal ions Hg preferentially binds with sulphur and nitrogen rich ligands (amino acids) and entered inside the cells. Hg effect damage include blocking functional groups of enzymes, polynucleotide’s, or transport systems for nutrient ions, denaturing and inactivating enzymes and disrupting cell and organelle membrane integrity (Ochiai, 1987). The possible causal mechanisms of Hg toxicity are changes in the permeability of the cell membrane, reactions of sulphydryl (-SH) groups with cations, affinity for reacting with phosphate groups and active groups of ADP or ATP and replacement of major cations (Kabata-Pendias and Pendias, 1989).
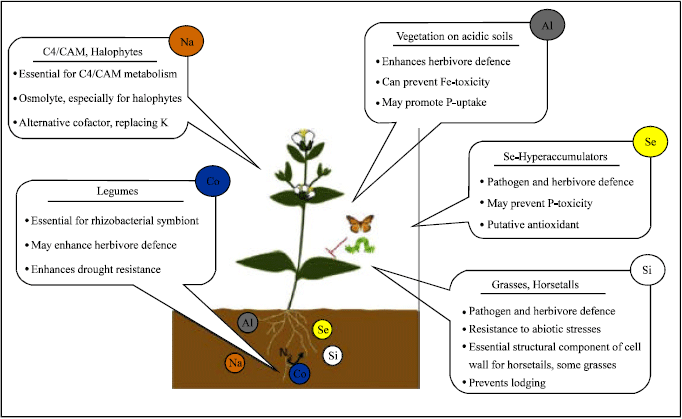 | |
| Fig. 1: | The mechanisms responsible for the growth promoting effects of the five beneficial elements Al, Co, Na, Se and Si (Pilon-Smits et al., 2009) |
Heavy metals like copper (Cu) and iron (Fe) can be toxic in excess amount because of their participation in redox cycles producing hydroxyl radicals which are extremely toxic to living cells (Stohs and Bagchi, 1995). Unlike Cu and Fe, Cd is a non-redox metal that is strongly phytotoxic and caused growth inhibition and plant death. Cd induced changes in lipid profile (Ouariti et al., 1997) and by affecting the enzymatic activities associated with membranes, such as the H+- ATPase (Fodor et al., 1995). Cd is also reported to damage the photosynthetic apparatus (Siedlecka and Baszynsky, 1993), decrease chlorophyll content and inhibit the stomatal regulations (Barcelo and Poschenrieder, 1990). The major storage site for Zn and Cd in plants is cell wall of roots, vacuoles of epidermis and bundle sheath of leaves (Hu et al., 2009). The Cd influx and efflux in leaf mesophyll layer depends on the expression of plasma membrane and tonoplast transporters. Root metal uptake rates are increased, generally through an increase in VMAX, without major changes in KM, of root metal uptake rates (Lasat et al., 1996; Lombi et al., 2001).
Works on Alyssum lesbiacum has given a base of Nickel uptake into vacuoles from leaf tissue and this was enhanced by the presence of Mg/ATP, presumably via energisation of the vacuolar H+-ATPase (Ingle and Fricker, 2008). The model hyperaccumulator Thlaspi caerulescens is capable to tolerat Zn, Cd and Ni and it has been shown that part of the Ni is translocated as a stable Ni-NA complex in the xylem sap (Mari et al., 2006).
BIOMASS PRODUCTION
The discussion of the effect of heavy metal on biomass productions of metal hyperaccumulation is restricted here to Cd, Pb, Zn, V and Cr.
In metal hyperaccumulators the biomass production level depends on the concentration of the metals and duration of exposures e.g., The biomass is negatively correlate with Cd concentration in B. napus, cultured in the nutrient solution and the reduction in dry weight was significantly higher for the root than the shoot. Considering the high aboveground biomass production and Cd accumulation in the shoot, B. napus can be a potential candidate for the phytoextraction of Cd (Selvam and Wong, 2008).
Biomass production were extensively studied in split pots filled with soil spiked at 0, 3, 6, 12, 25 and 50 mg Cd kg-1 soil by Selvam and Wong (2009) and revealed decline in biomass production which established that cadmium is toxic for biomass production.
Suitable levels of Pb, Zn and Cd could stimulate the biomass production in A. paniculata and thus, it provides a new plant material for understanding the mechanisms of stimulatory effect and co-hyperaccumulation of multiple heavy metal (Tang et al., 2009). However, biomass production is inhibited in Glycine max and Phaseolus vulgaris in treated with VOSO4 (Kaplan et al., 1990). Similarly in E. fluctuans, biomass production was not affected up to concentration of 2.5 mg L-1 V2O5 when exposed for 7 days (Fig. 2). However, V2O5 concentration >2.5 mg L-1 significantly reduced the biomass and increased in treatment duration enhanced the V2O5 toxicity (Sarma et al., 2009). The author recorded that after 21 days exposure of 10.0 mg L-1 V2O5 to E. fluctuans approximately 42.47% biomass was decreased (Fig. 2). Similarly in Vallisneria spiralis L., an increased in treatment duration enhanced the chromium toxicity and 0.1 μg mL-1 Cr caused 7% decrease in biomass after 48 h and 64% loss of biomass was recorded after 72 h exposure of 10 μg mL-1 Cr (Vajpayee et al., 2001). The results addressed that heavy metals toxicity to biomass were concentration and duration’s dependent.
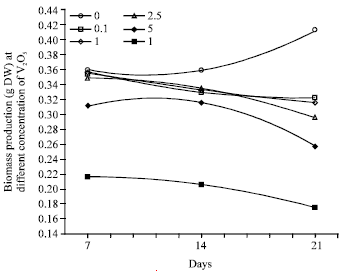 | |
| Fig. 2: | Biomass production of vanadium treated E. fluctuant was negatively correlated with concentration of vanadium in nutrient medium with increased in duration |
However, modern transgenic research approach at present going on for introduction of novel traits into high biomass plants for development of effective phytoremediation technologies. A number of transgenic high biomass yield plants have been successfully generated in an attempt to modify the tolerance, uptake or homeostasis of trace elements (Kramer and Chardonnens, 2001).
BIOCHEMICAL RESPONSES
Several heavy metals such as Fe, Cu, Co, Mn, Mo and Ni are essential elements to plant metabolism. In higher concentrations, many heavy metals inhibit plants biochemical production and this has been extensively studied and reviewed (Fernandes and Henriques, 1991; Sarma and Sarma, 2007; Sarma et al., 2009). Photosynthetic pigments of plants belonging to different group’s exhibit differential tolerance to metals (Vajpayee et al., 2001). Heavy metal substituted chlorophylls and related porphyrins have been known in vitro for a long time (Kupper et al., 2000). Many researchers examined the effect of heavy metals on photosynthesis and observed a decrease in fluorescence (Atal et al., 1991; El-Sheekh, 1992). Cd induced reduction of photosynthetic pigment were recorded in two species viz., M. heterophyllum and P. crispus (Fig. 3a, b and 4a, b). The highest decrease in chlorophyll a was recorded in 7.34 mg g-1 in M. heterophyllum and 8.09 mg g-1 in P. crispus (at 64 mg L-1 and 96 h) have given as evidence for the Cd toxicity to chlorophyll.
Cadmium hyperaccumulator Atriplex halimus subsp. schweinfurthii was sensitive to high Cd results reduction of chlorophyll pigments, stomatal transpiration and root hydraulic conductivity (Nedjimi and Daoud, 2009). The Cr uptake by many aquatic plants influenced in biochemical process results alteration of pigments and amino acids. It has been reported that Cr (VI) causes toxicity to δ-amionolevulinic acid dehydratase (an enzyme involved in Chlorophyll biosynthesis) by impairing δ-amiono levulinic acid (ALA) utilizations (Vajpayee et al., 2000). Further, Vajpayee et al. (2000) suggested that Cr (VI) could exchange the Mg from active site of enzyme resulting into phaeophytin and thus depleted chlorophyll contents in Cr treated plants. Chromium also inhibits chlorophyll biosynthesis by creating nutrient imbalance (Barcelo et al., 1986). It has been reported that chromium induced degradation of carotenoid in some plants while in contrast Vajpayee et al. (2001) reported that carotenoid contents was increased in Cr treated V. spiralis. Thus, the effects of heavy metals on carotenoid contents were plants and metal specific.
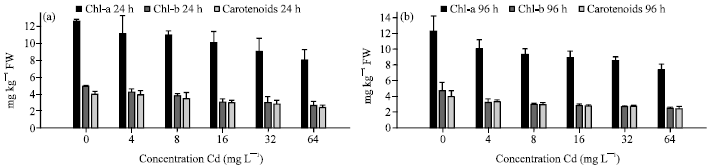 | |
| Fig. 3: | Chlorophyll a, b and carotenoid contents of (mg g-1 FW) M. heterophyllum exposed to various Cd concentrations for (a) 24 and (b) 96 h (Sivaci et al., 2008) |
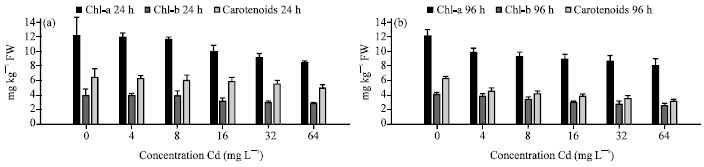 | |
| Fig. 4: | Chlorophyll a, b and carotenoid contents of (mg g-1 FW) P. crispus exposed to various Cd concentrations for (a) 24 and (b) 96 h (Sivaci et al., 2008) |
Mercuric cations have a high affinity for sulphydryi (-SH). In almost all proteins contain sulphydryl groups or disulphide bridges, Hg could disturb the normal functions of proteins in binding in two sites of a protein molecule without deforming the chain, lead to protein precipitation. (Clarkson, 1972).
Mercury affects both light and dark reactions in photosynthesis and caused inhibitions of electron transport activity, oxygen evolution and quenching of chlorophyll fluorescence in photosystem II (PS II). Substitution of the central atom of chlorophyll, magnesium, by mercury in vivo is an important damage mechanism, because it prevents photosynthetic light harvesting in the affected chlorophyll molecules and results in the breakdown of photosynthesis (Krupa and Baszynski, 1995).
GENETIC AND MOLECULAR BASIS OF METAL HYPERACCUMULATION
Metal hyperaccumulation is a fascinating phenomenon, which has interested scientists for over a century. Hyperaccumulators constitute a group of exceptional plant species and they possesses genetically inherited traits of metals hyperaccumulation and tolerance. The understanding of metal hyperaccumulation physiology has recently improved as a result of the development of molecular tools (Verbruggen et al., 2009). Transgenic approaches successfully employed to promote phytoextraction of metals (Cd, Pb and Cu) and metalloids (As, Se) from contaminated soil by their accumulation in the aboveground biomass involved mainly implementation of metal transporters, improved production of enzymes of sulphur metabolism and production of metal-detoxifying chelators metallothioneins and phytochelatins (Kotrba and Najmanova, 2009).
Recent research revealed that Arabidopsis thaliana has eight genes encoding members of the type 1 B heavy metal transporting subfamily of the P-type ATPases. Three of these transporters, HMA2, HMA3 and HMA4, are closely related to each other and are most similar in sequence to the divalent heavy metal cation transporters of prokaryotes (Hussain et al., 2004).
Quantitative mRNA in situ hybridization (QISH) in Thlaspi caerulescens shows that transporter gene expression changes during cadmium (Cd)/zinc (Zn) hyperaccumulations. Members of the ZIP gene family, a novel metal transporter family first time identified in plants are capable of transporting a variety of cations including Cd, Fe, Mn and Zn. The different cellular expression patterns for ZNT1 and ZNT5 were recorded by Kupper and Kochian (2010) both belonging to the ZIP family of transition metal transporters. ZNT1 may function in micronutrient nutrition while ZNT5 may be involved in metal storage associated with hyperaccumulation. Cadmium induced changes in cellular expression for ZNT1, ZNT5 and MTP1 could also be part of plants acclimatization to Cd toxicity.
The integral membrane protein Thlaspi goesingense metal tolerance protein 1 (TgMTP1) has been suggested to play an important role in Zn hyperaccumulation. High levels of TgMTP1 at the vacuolar membrane in shoot tissue of the Zn hyperaccumulator T. goesingense has been played an important role in both Zn tolerance and enhanced Zn uptake and accumulation, via the activation of a systemic Zn deficiency response (Gustin et al., 2009).
An antiporter is an integral membrane protein which is involved in secondary active transport of two or more ions across a phospolipid membrane in opposite directions. A recent genomic analysis provides a breakthrough in acquisition of zinc hypertolerance and hyperaccumulation characters via involvement of Zn2+/H+ antiporter. Metal Tolerance Protein 1 (MTP1) is one of genes present in A. halleri which encodes a Zn2+/H+ antiporter involved in cytoplasmic zinc detoxification and developed a zinc tolerance characteristic (Shahzad et al., 2010).
Plant tissue cultures such as callus, cell suspensions and hairy roots are extensively used in phytoremediation research as model plant systems. Many studies have demonstrated that plant tissue cultures are an extremely valuable tool in phytoremediation research. The results derived from tissue cultures can be used to predict the responses of plants to environmental contaminants and to improve the design and thus reduce the cost of subsequent conventional whole plant experiments (Doran, 2009).
ANTIOXIDANTS DEFENSE SYSTEM IN METAL HYPERTOLERANCE
Phytochelatins are oligomers of glutathione, produced by the enzyme phytochelatin synthase. They are found in plants, fungi, nematodes and all groups of algae including cyanobacteria. Phytochelatins act as chelators and are important for heavy metal detoxification (Ha et al., 1999). They are abbreviated PC2 through PC11. Plants possess several antioxidative defense systems to scavenge toxic free radicals in order to protect themselves from the oxidant stress including that caused by heavy metals. The antioxidative defense system falls into two general classes: (1) low molecular weight antioxidants, which consist of lipid-soluble membrane-associated antioxidants (e.g., α-tocopherol and β-carotene) and water-soluble reductants (e.g., glutathione and ascorbate) and (2) antioxidative enzymes: Superoxide Dismutase (SOD) Ascorbate Peroxidase (APX), Catalase (CAT) and Glutathione Reductase (GR).
Glutathione (GSH), a sulfur containing tripeptide, is considered to be the most important cellular antioxidant involved in cellular defense against toxicants (Scott et al., 1993) and function directly as a free radical scavenger. This antioxidant is also the precursor for the phytochelatins that act as heavy metal binding peptides in plants (Rosen, 2002). GSH levels in plants are known to change under metal stress (Koricheva et al., 1997).
The Cd treatments significantly enhanced the synthesis of phytochelatins (PCS) in plants. However, Sun et al. (2010) reported that the variation in phytochelatins productions in root and shoots in two Cd treated species viz., R. globosa and R. islandica may be used as a biomarker of Cd hyperaccumulation and the synthesis of PCS may be related to an increase in the uptake of Cd ions into the cytoplasm, not the primary mechanism for Cd tolerance. Similarly the accumulation of Cd has influenced on the synthesis of phytochelatins in Brassica napus and in the shoot, the concentration of PC3 and PC4 was higher than the PC2 irrespective of the quantity of Cd uptake (Selvam and Wong, 2008). This result gives an evidences that the detoxification of Cd involves higher molecular weight thiol complexes in the shoot. In Arabis paniculata after Cd exposure induced formation of PCS and three unknown thiols in the roots, but none were detected in the shoots (Zeng et al., 2009). Sesbania sp. responded to Hg induced oxidative stress by modulating non-enzymatic antioxidants viz., Glutathione (GSH) and Non-Protein Thiols (NPSH) and enzymatic antioxidants: Superoxide Dismutase (SOD), ascorbate peroxidase (APX) and Glutathione Reductase (GR). The Sesbania plants were able to tolerate Hg induced stress using effective antioxidative defense mechanisms (Israr et al., 2006; Israr and Sahi, 2006). It has been suggested that phytochelatins play a constitutive role in plant metal tolerance (Zenk, 1996). However, evidence provided by Zenk (1996) for this role is not conclusive and could just as easily indicate a stress response (e.g., production of phytochelatins upon exposure to metals).
Phytoremediation is initiated all over the globe and this has considered one of the low-cost novel green technologies. The physiological and molecular basis of metal hyperaccumulation in plants are still in research and development phase. This review has focused on recent evidence that identifies potential molecular mechanism that may be involved in the resistance, tolerance as well as hyperaccumulation of heavy metals. The findings suggest that in some plants ZIP family genes contribute to metal hyperaccumulation and transport, but their individual functions yet to be identified and further intensive research is needed in this concern. The identifications of individual functions of metal transporters will help to develop the knowledge of plants metal homeostasis. Results already obtained have been indicated that the physiological and biochemical responses were plants and metal specific. Identification of novel genes with high biomass yield characteristics and the subsequent development of transgenic plants with superior remediation capacities will be encouraging further research. In depth research study is warranted to find out which plant is maximum resistant and best adapted in particular metallic environment or region. In situ toxicity text could be beneficial for initial identification of particular species.
I am thankful to all colleagues, in particular to current and previous members of my research team, for their supports. I thank to all my Lab mates of Ecosystem Research Laboratory, North-Eastern Hill University. I am grateful to Ms. Idaiarilin Lyndogh Synrem for reading the fist draft of the manuscripts and Mr. Souravjyoti Borah for technical assistant. Funding by a Dr. D.S. Kothari Post Doctoral Fellowship from the University Grants Commission, New Delhi (F. 4-2/2006 (BSR)/13-177/2008 (BSR) is gratefully acknowledged. Finally, I apologize to all colleagues for those parts of their work that were not cited in the manuscripts because of restrictions in space.