Research Article
Biological Treatment of Tannery Wastewater - A Review
Department of Chemical Engineering, Annamalai University,Annamalainagar-608002, Tamilnadu, India
M. Rajasimman
Department of Chemical Engineering, Annamalai University,Annamalainagar-608002, Tamilnadu, India









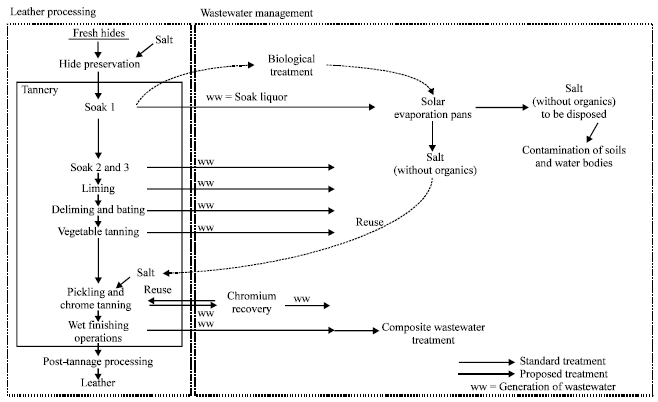
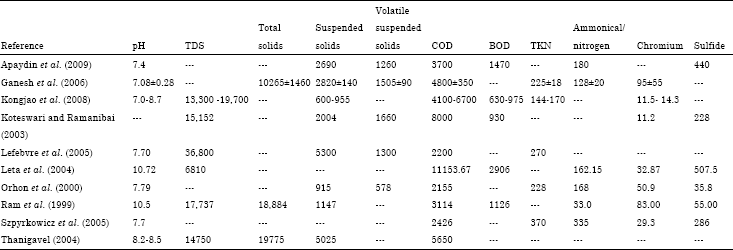
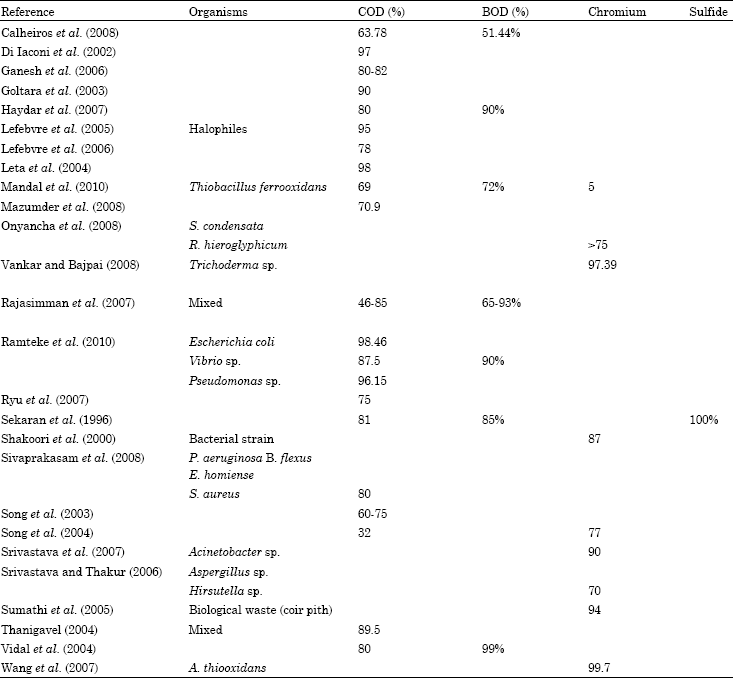
ashish kumar Reply
good paper
ashish kumar Reply
good review