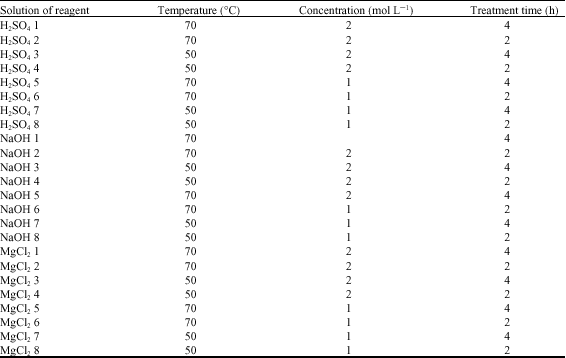
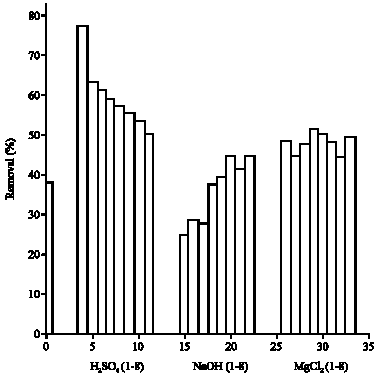
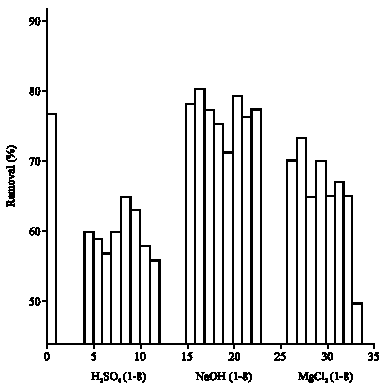
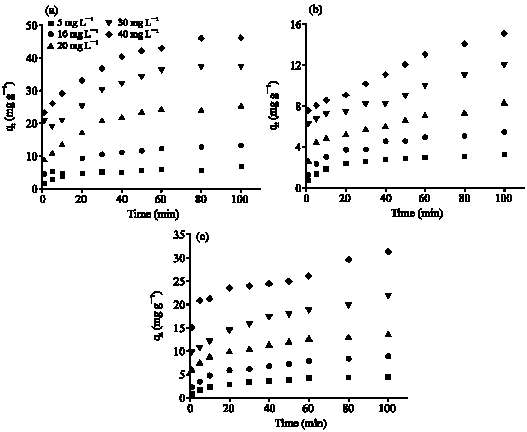
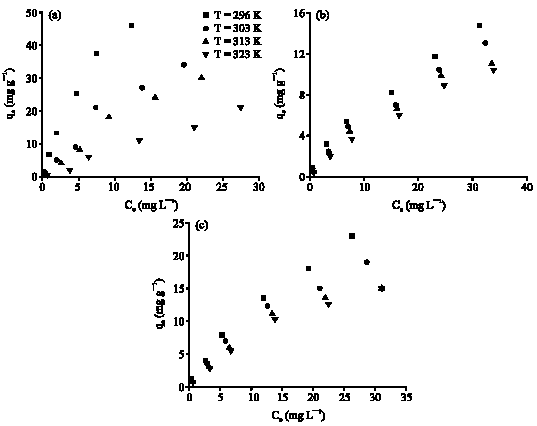
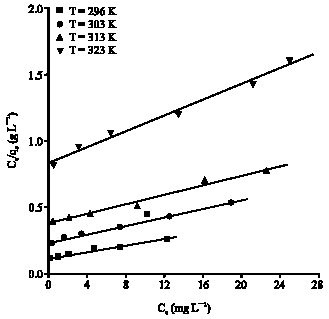
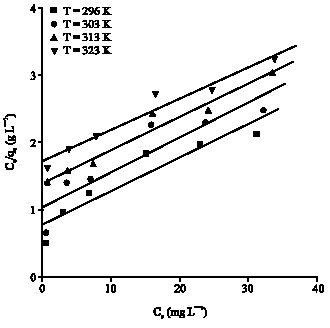
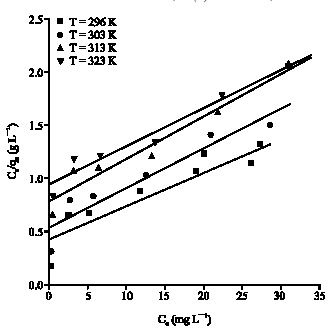
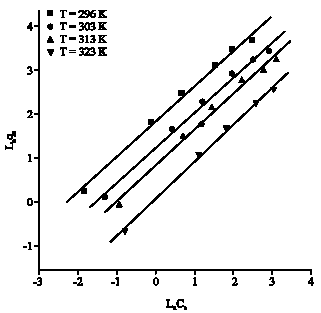
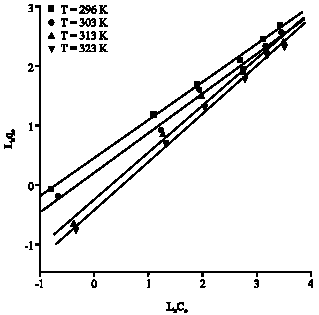
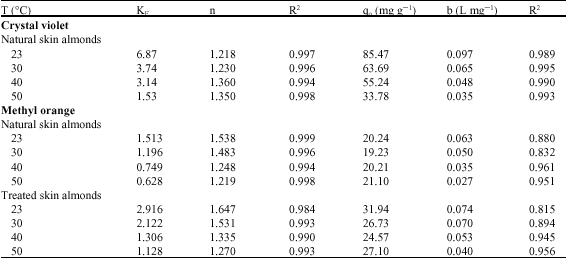
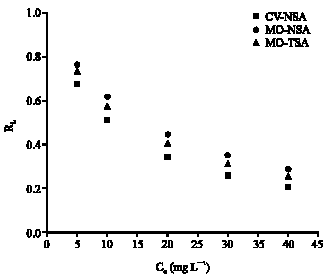
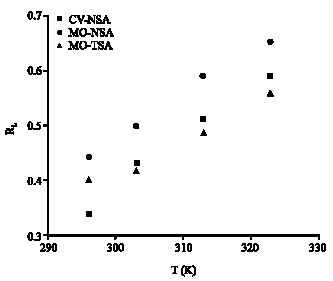
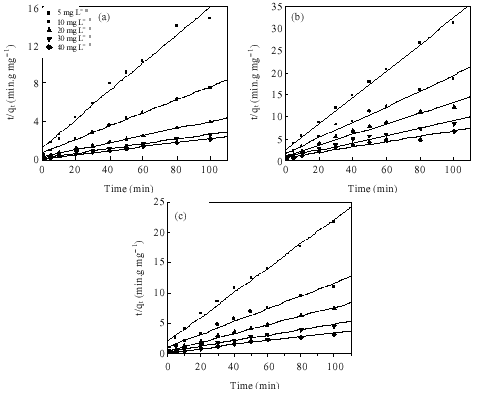
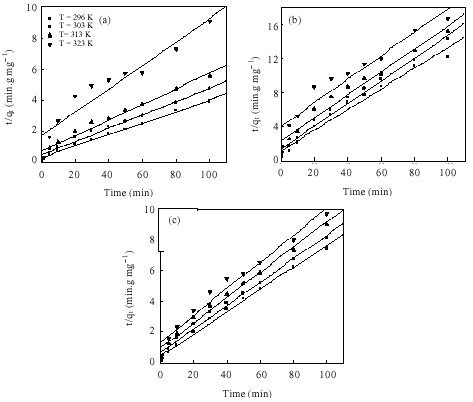
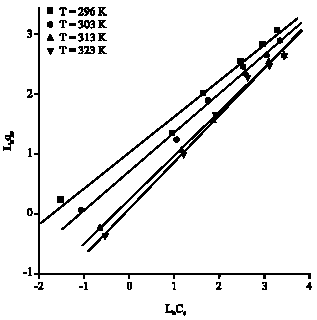Research Article
Synthetic Textile Effluent Removal by Skin Almonds Waste
Laboratoire de Genie de la Reaction, Faculte de Genie Mecanique et Genie des Procedes, Universite des Sciences et de la Technologie Houari Boumediene, BP 32 El-Alia Bab Ezzouar, Alger, Algerie
A. Bensmaili
Laboratoire de Genie de la Reaction, Faculte de Genie Mecanique et Genie des Procedes, Universite des Sciences et de la Technologie Houari Boumediene, BP 32 El-Alia Bab Ezzouar, Alger, Algerie
N.Y. Mezenner
Laboratoire de Genie de la Reaction, Faculte de Genie Mecanique et Genie des Procedes, Universite des Sciences et de la Technologie Houari Boumediene, BP 32 El-Alia Bab Ezzouar, Alger, Algerie