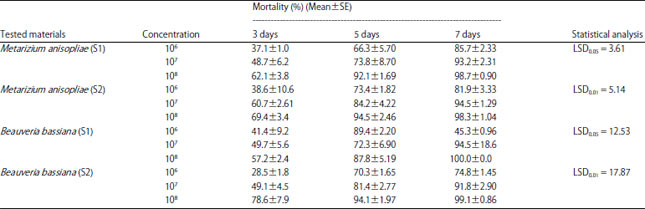
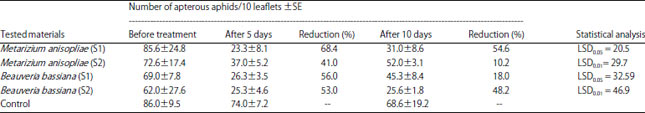
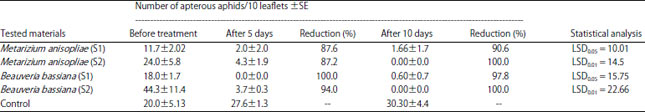
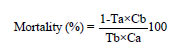Research Article
Effectiveness of Two Entomopathogenic Fungi Sources Toward Some Sucking Insects and Their Predators on Okra Crop
Department of Pests and Plant Protection, National Research Centre, Cairo, Egypt
LiveDNA: 20.16789
Sawsan S. Moawad
Department of Pests and Plant Protection, National Research Centre, Cairo, Egypt
LiveDNA: 20.16594
Hanaa E. Sadek
Department of Pests and Plant Protection, National Research Centre, Cairo, Egypt
LiveDNA: 20.16791
Dalia E. Lotfy
Plant Protection Research Institute, Agriculture Research Centre, Cairo, Egypt