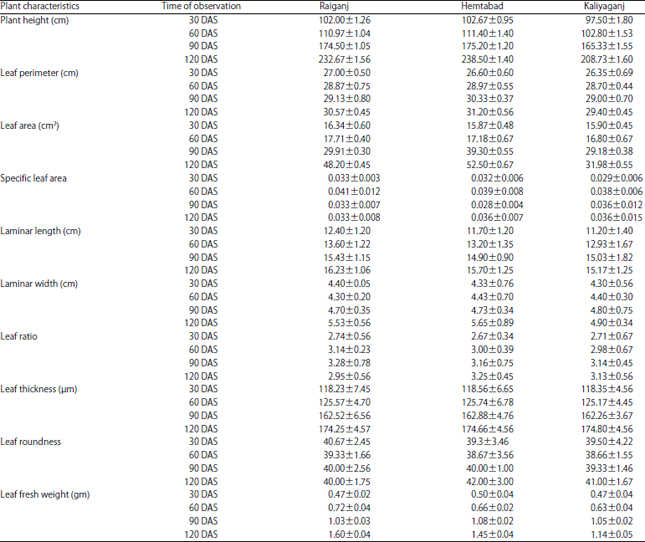
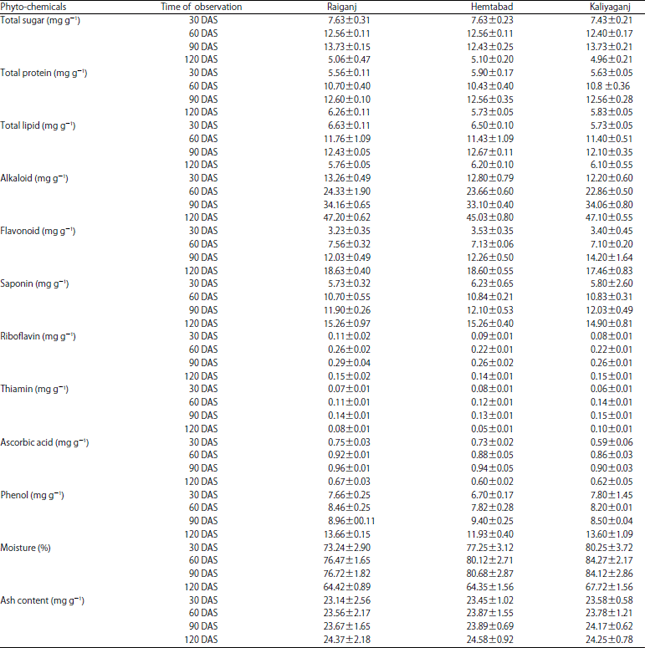
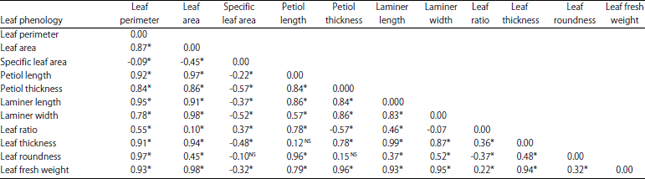
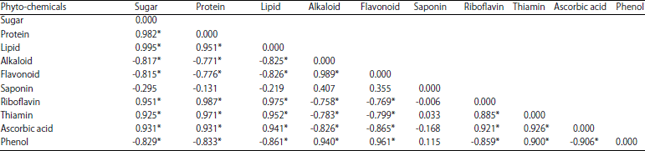
Research Article
Defensive Response of Corchorus olitorius in Relation to Infestation of Anomis sabulifera
Entomology Laboratory, Department of Zoology, University of Gour Banga, Malda, 732103, West Bengal, India
LiveDNA: 91.16420
Shubham Bhattacharyya
Immunopharmacology and Molecular Cell Biology Laboratory, Department of Zoology, University of Gour Banga, 732103 Malda, West Bengal, India
Subhrajyoti Roy
Immunopharmacology and Molecular Cell Biology Laboratory, Department of Zoology, University of Gour Banga, 732103 Malda, West Bengal, India
Kaushik Chakraborty
Entomology Laboratory, Department of Zoology, University of Gour Banga, Malda, 732103, West Bengal, India