Research Article
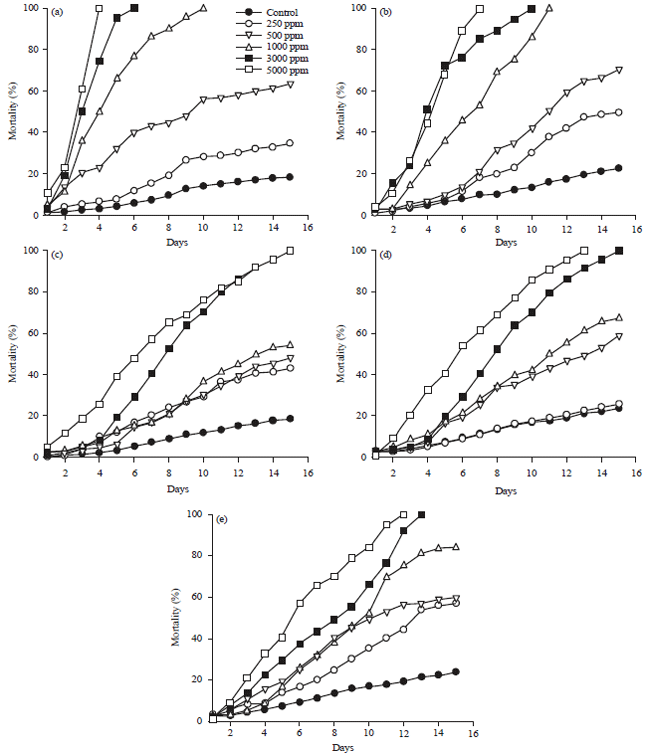
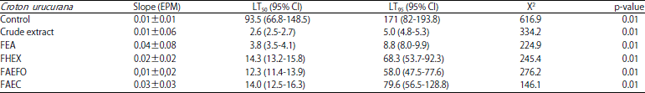
Toxicity of Croton urucurana Against the Subterranean Termite Heterotermes sulcatus (Isoptera: Rhinotermitidae)
Programa de Pos-graduacao em Agronomia-Fitotecnia, Universidade Federal do Piaui, Campus Profa, Cinobelina Elvas, Bom Jesus-PI, 64900-000, Brazil
Angelica Silva Oliveira
Programa de Pos-graduacao em Agronomia-Fitotecnia, Universidade Federal do Piaui, Campus Profa, Cinobelina Elvas, Bom Jesus-PI, 64900-000, Brazil
Gabriel dos Santos Carvalho
Programa de Pos-graduacao em Agronomia-Fitotecnia, Universidade Federal do Piaui, Campus Profa, Cinobelina Elvas, Bom Jesus-PI, 64900-000, Brazil
Leonardo Santana da Silva
Universidade Federal de Sao Paulo, UNIFESP, Campus Diadema, Rua Artur Riedel, 275, Eldorado, Diadema-SP, 09972-270, Brazil
Luciana Barboza Silva
Programa de Pos-graduacao em Agronomia-Fitotecnia, Universidade Federal do Piaui, Campus Profa, Cinobelina Elvas, Bom Jesus-PI, 64900-000, Brazil
Eliane Carneiro Bueno dos Santos
Programa de Pos-graduacao em Agronomia-Fitotecnia, Universidade Federal do Piaui, Campus Profa, Cinobelina Elvas, Bom Jesus-PI, 64900-000, Brazil
Fabiana Elaine Casarin
Universidade Federal de Sao Paulo, UNIFESP, Campus Diadema, Rua Artur Riedel, 275, Eldorado, Diadema-SP, 09972-270, Brazil