Research Article
Impact of Arid Land Conditions on Biological Activities of Honeybee Colonies
Department of Plant Protection and Biomolecular Diagnosis, Arid Lands Cultivation Research Institute, City of Scientific Research and Technological Applications (SRTA-City), New Borg El-Arab, 21934 Alexandria, Egypt
A.A.A. Abdelaal
Department of Economic Entomology and Agricultural Zoology, Faculty of Agriculture, Minuofia University, Shebin Elkom, Egypt










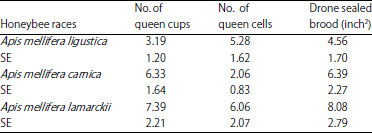

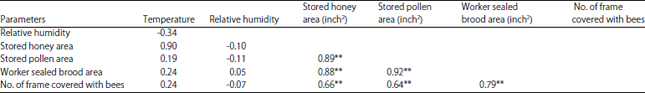
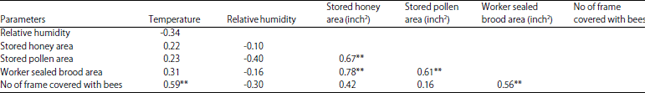
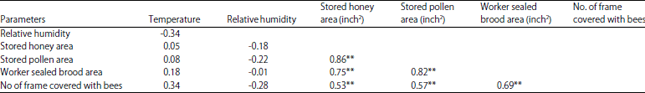
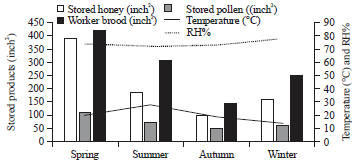
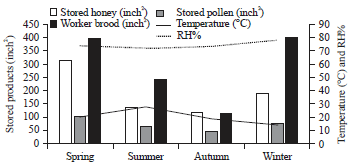
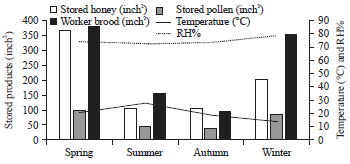
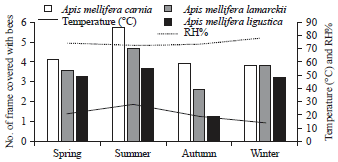
rami Issa Reply
i want this paper iam researcher