Research Article
Influence of Prey Types on the Biological Characteristics of Cosmolaelaps qassimensis (Acarai: Laelapidae)
Department of Agricultural Zoology, Faculty of Agriculture, Mansoura University, Egypt
N.F. Abdel-Baky
The food sources for different groups of soil mites include nematodes, arthropods and plant roots (Inserra and Davis, 1983; Zaher, 1986). Their numbers could be related to the abundance of their food sources. Some of soil mites are effective predators capable of regulating and suppressing populations of their prey including plant parasitic nematodes (Al Rehiayani and Fouly, 2005; Mostafa et al., 2013).
Beneficial soil mites, especially predatory species, must be conserved in the field to promote a more stabilized pest and natural balance (Abo-Taka et al., 1992; Lobbes and Schotten, 1980). Among the predatory mites, the mesostigmata represent an important component of the below ground food web, where they are generally considered to be predators feeding on small arthropods, worms and nematodes. Several predatory mites such as Hypoaspis near aculeifer (Inserra and Davis, 1983), Cosmolaelaps simplex (Berlese) (Al Rehiayani and Fouly, 2005), Lasioseius scapulatus (Kennet) (Imbriani and Mankau, 1983), Lasioseius dentatus (Fox) (Fouly, 1997) and Lasioseius athiasae Nawar and Nasr (Abou-Awad et al., 2001) had the ability to capture, consume and complete its entire life cycle on nematode species.
Cosmolaelaps (Berlese) consists of a large group of free-living predators in soil. The laelapidae mite, Cosmolaelaps qassimensis is a new species which was recently recorded and identified by Fouly and Al-Rehyiani (2014), where its biology and life history has never been studied so far.
The objective of the present study was to determine different biological characteristics of C. qassimensis fed on the acarid mites, Tyrophagus putrescentiae (Schrank) and Caloglyphus rodriguez Samsinak in comparison with the egg masses of the root-knot nematode, Meloidogyne incognita Chitwood under laboratory conditions.
Acarid mite and nematode cultures: Stock cultures of both acarid mite species were maintained in laboratory, provided with on mould cheese and pollen as food sources. Egg masses of root-knot nematode were provided from a culture of M. incognita maintained on tomato plants in the greenhouse of Nematology Branch, Faculty of Agriculture and Veterinary Medicine, Qassim University in 2013.
Culture of C. qassimensis and experiment technique: Laboratory culture of C. qassimensis was originated from soil samples under date palm trees at Qassim region, Saudi Arabia in 2013. Mite samples have been maintained on the acarid mite species, T. putrescentiae and pollen of date trees as food sources in rearing units which was previously described by Fouly (1996). Newly deposited eggs of C. qassimensis had been singly placed in smaller rearing units (5 cm in diameter) using a fine brush. Immature stages and adults of the predator were provided with one of the tested food types (the acarid mites, T. putrescentiae and C. rodriguez) in addition to the egg masses of the root-knot nematode, M. incognita, during their entire life span. All experimental units were kept in an incubator at 26±1°C and 70±5% RH. In all cases, incubation period, duration of each stage (in days), number of surviving laelapidae mite individuals and egg production were recorded daily.
Statistical analysis: Data of incubation and developmental time, duration of adult male and female, female reproductive period as well as fecundity were analyzed using one-way ANOVA followed by the LSD test of Duncan multiple range test to compare means by using Costat Software (1990).
Life table parameters: Life table parameters of the predatory mite C. qassimensis which was fed on three different prey species, were calculated according to Birch (1948) and Laing (1968) by using the basic computer program Abou-Setta et al. (1986) where, L is number of alive females, x is actual female age (time from egg stage till death) (in days), Mx is number of female progeny/female (mothers)/day x (specific fecundity rate), Lx is rate of survival at day x, Ro net reproductive rate (ΣLxMx), T mean generation time (ΣLxMx.x)/(ΣLxMx), DT is time required for the population to double its number, GRR is Gross Reproductive Rate, rm intrinsic rate of natural increase, erm finite rate of increase. Moreover, the Doubling Time (DT) was determined according to Laughlin (1965) and Carey (1993).
Duration of development and fecundity of C. qassimensis: Developmental stages and adults of C. qassimensis were fed on the three preys T. putrescentiae, C. rodriguez and egg masses of M. incognita during their whole life span and incubated at 26±1°C and 70±5% RH. Results showed that there was no significant difference between the incubation period of eggs for both females (F = 3.77 and p = 0.029) and males (F = 3.71 and 0.033) (Table 1, 2). The durations of larva, protonymph, deutonymph and life span of C. qassimensis females were statistically sub-equal when they fed T. putrescentiae and C. rodriguez while they were significantly shorter than those on M. incognita (F = 18.19, 7.17 and 18.06 for larvae, protonymph and deutonymph, respectively (p<0.01) (Table 1). Total immature stages, life cycle, pre-oviposition, oviposition and post-oviposition periods of C. qassimensis were significantly shorter when predatory mites fed on T. putrescentiae and followed by C. rodriguez and egg masses of M. incognita, respectively (F = 37.99, 32.52, 94.98, 132.6 and 36.78, respectively and p<0.01). As shown in Table 2, female longevity of C. qassimensis did not significantly differ according to prey species (F = 0.24 and p = 0.785).
Durations of larva, protonymph, total immature development, life cycle, longevity and life span of C. qassimensis males were significantly shorter when fed on T. putrescentiae in compared with C. rodriguez and M. incognita (F = 17.16, 11.32, 49.73, 45.02, 87.95 and 102.80, respectively, p<0.01). The deutonymph took the same period when it fed on either T. putrescentiae or C. rodriguez, while feeding on egg masses of M. incognita, caused the longest duration before reaching maturity (F = 13.20 and p<0.01) (Table 2).
The total numbers of deposited eggs by each female mite was significantly higher for female fed T. putrescentiae and followed by C. rodriguez and then egg masses of M. incognita, which occupied the last rank (F = 135.0 and p<0.01) (Fig. 1).
| Table 1: | Duration (in days) of Cosmolaelaps qassimensis female developmental stages fed on Tyrophagus putrescentiae, Caloglyphus rodriguez and egg masses of Meloidogyne incognita and incubated at 26±1°C and 70±5% RH |
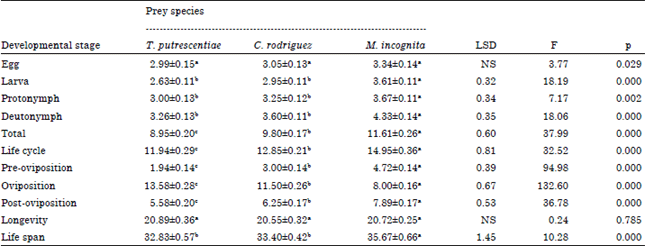 | |
| Values represented by Mean±SE. Means have the same letter in each row haven’t significant differences (p = 0.05) by Duncan multiple range test, NS: No significant differences where df = 2, 54 | |
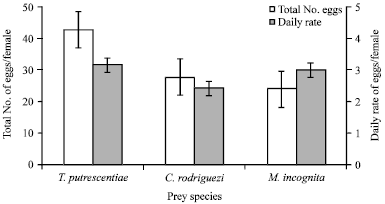 | |
| Fig. 1: | Total and daily rate of deposited eggs/female of Cosmolaelaps qassimensis fed Tyrophagus putrescentiae, Caloglyphus rodriguez and egg masses of Meloidogyne incognita and incubated at 26±1°C and 70±5% RH |
| Table 2: | Duration (in days) of Cosmolaelaps qassimensis male developmental stages fed on Tyrophagus putrescentiae, Caloglyphus rodriguez and egg masses of Meloidogyne incognita and incubated at 26±1°C and 70±5% RH |
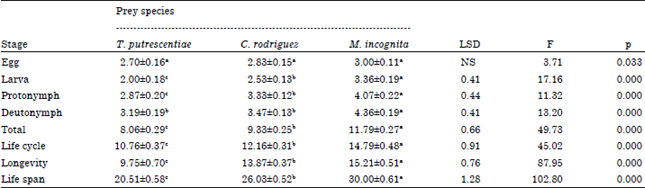 | |
| Values represented by Mean±SE. Means have the same letter in each row haven’t significant differences (p = 0.05) by Duncan multiple range test, NS: No significant differences where df = 2 and 42 | |
Also, the daily rate of eggs per female was higher on T. putrescentiae (3.14 eggs) followed by egg masses of M. incognita (2.98 eggs/female/day) and was lowest on C. rodriguez (2.41 eggs/female/day) (Fig. 1).
Life table parameters of C. qassimensis: Data represented in Table 3 clarified that sex ratio and survival (%) of C. qassimensis were at their highest values, when the predatory mites fed T. putrescentiae and followed by C. rodriguez and M. incognita. While, mean generation Time (T) and the time required for C. qassimensis population to double its numbers (DT) were shorter when it fed T. putrescentiae in comparison with C. rodriguez and M. incognita. With regard to the survivorship of the laelapidae mite, C. qassimensis, Lx curves followed the I pattern in which survival of immature stages was 92, 90 and 86% when fed on T. putrescentiae, C. rodriguez and egg masses of M. incognita, respectively. Death of C. qassimensis females started after the longest period of oviposition (13 days) on T. putrescentiae, while by feeding on C. rodriguez and egg masses of M. incognita, female death started after 9 days of oviposition (Fig. 2). As shown in Fig. 3, the age specific fecundity (Mx) of C. qassimensis was clearly affected by prey species. Moreover, results showed that the net reproductive rate (Ro) of C. qassimensis reached its highest rate of 30.99 female egg/female/day, when it was provided with the acrid mite T. putrescentiae.
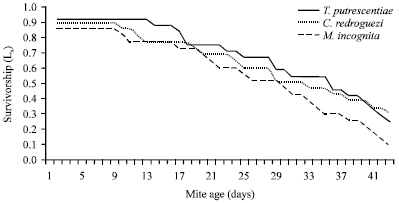 | |
| Fig. 2: | Survivorship (Lx) of Cosmolaelaps qassimensis fed on Tyrophagus putrescentiae, Caloglyphus rodriguez and egg masses of Meloidogyne incognita at 26±1°C and 70±5% RH |
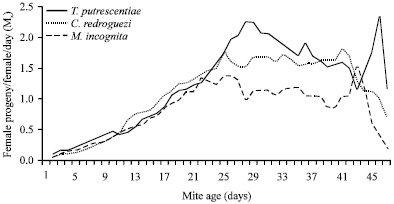 | |
| Fig. 3: | Age specific fecundity (Mx) of Cosmolaelaps qassimensis fed on Tyrophagus putrescentiae, Caloglyphus rodriguez and egg masses of Meloidogyne incognita at 26±1°C and 70±5% RH |
| Table 3: | Life table parameters of Cosmolaelaps qassimensis fed on Tyrophagus putrescentiae, Caloglyphus rodriguez and egg masses of Meloidogyne incognita at 26±1°C and 70±5% RH |
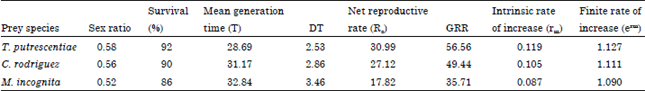 | |
| Survival (%): Percentage of immature stages reached maturity, GRR: Gross reproductive rate, DT: Time required for the population to double its number | |
This value clearly decreased to 27.12, when the laelapidae mite fed on C. rodriguez, while eggs of nematode occupied the last rank in food preference with only 17.82 eggs/female/day (Table 3). Similar trend was noticed with the intrinsic rate of natural increase (rm) and finite rate of increase (erm), where T. putrescentiae proved to be the most preferable prey and followed by C. rodriguez, while nematode eggs came last.
Biology of C. qassimensis: The incubation period of C. qassimensis eggs was not affected by prey species. These findings agree with those of Fouly (1997) who reared the ascid mite L. dentatus on both acarid mites T. putrescentiae and C. rodriguez in addition to the green mould fungus Penicillium digitatum Sacc. and egg masses of root-knot nematode, M. javanica Treub. Also, Abou-Awad et al. (2001) mentioned that the incubation period of the ascid mite, L. athiasae Nawar and Nasr did not changed by prey species. Moreover, Al Rehiayani and Fouly (2005) found similar results when C. simplex (Berlese) fed acarid mites and nematodes.
Both sexes of C. qassimensis passed through larval, protonymphal and deutonymphal stages before reaching adulthood. Larvae did not feed on any of the tested prey species. Similar results were previously obtained with Hypoaspis vacua (Micheal) (Abou-Awad et al., 1989), Pachylaelaps aegyptiaca Hafez and Nasr (Gomaa et al., 1989), C. claviger (Berlese) (Affifi and Van der Geest, 1991), L. athiasae (Abou-Awad et al., 2001), who reported that larval feeding did not seem to be necessary for successful development. Contradictory, it was mentioned before that larval stage of most mesostigmatid mites must feed to develop into protonymphs (Osman et al., 1988; Walter and Lindquist, 1989; Fouly et al., 1994, 1995; Fouly, 1997; Abou-Setta et al., 1997). The previous data indicate that a diet of egg masses of the plant parasitic nematode, M. incognita obviously prolonged the developmental periods of both sexes of C. qassimensis in comparison with the acarid mites. Data also showed that there were significant differences between the effects of both acarid mite species as preys on the developmental period of the tested predaceous mite. Freira and de Moraes (2007) found that C. paulista Freire and Moraes took only 8.6 days to develop from egg to adult when it was provided with T. putrescentiae and kept at 26°C. From the previous results, it can be concluded that C. qassimensis can be considered as a polyphagous predatory mite which can survive and reproduce on a wide range of prey species. These results agree with those of Al Rehiayani and Fouly (2005) who found that C. simplex had been successfully reared on both acarid mite C. rodriguez and egg masses of two plant parasitic nematodes, M. javanica and Tylenchulus semipenetrans (Cobb). Also, Affifi and Van der Geest (1991) reported that C. claviger fed and reproduced when it was provided with the bulb mite, Rhizoglyphus echinopus, collembolan, eggs and larvae of house fly as well as free living nematodes.
Also, it was found that C. qassimensis reproductive potentiality was higher by feeding on mites or insects than on nematodes (Affifi and Van der Geest, 1991). Moreover, it was found that about 18 mesostigmatid mite species fed vigorously on the nematode species Cephalopus sp. (Rhabiditida) under laboratory conditions. Most of these species preferred the vermiform nematodes and showed also a positive response to the obese-form like females of Meloidogyne spp. and cyst nematodes Heterodera spp. that were sedentary (Muraoka and Nobuyoshi, 1976). Also, it was experimentally proved that L. dentatus survived and successfully reproduced by feeding on fungi, acarid mites and root-knot nematodes (Fouly, 1997). In contrast, Rodriguez (1964) found that the mite species Macrocheles muscadomestica (Scopoli) did not reproduce when it was supplied with acarid mites or collembolans but it had a successful biology on nematodes.
As in most mesostigmatid mites, C. qassimensis female lived for a longer time than male, (female lived for an average of 20.89, 20.55 and 20.72 days while males lived for 9.75, 13.87 and 15.21 days), when they were fed on T. putrescentiae, C. rodriguez and egg masses of M. incognita, respectively. Similarly, Al Rehiayani and Fouly (2005) reported that females of C. simplex lived for longer time (35.4, 33.3 and 34.7 days) than males (29.2, 33.2 and 34.3 days), when they were fed on C. rodriguez and both nematodes M. javanica and T. semipenetrans, respectively. Also, Ydergaard et al. (1997) mentioned that males of H. miles Berlese had a slight tendency to develop faster than females.
Our results showed that the total percentage of daughter females of C. qassimensis represented 58, 56 and 52% from the whole mite populations, when the predatory mite fed on T. putrescentiae, C. rodriguez and egg masses of M. incognita, respectively. These results are in agreement with those of Lobbes and Schotten (1980) and Enkegaard et al. (1997) who found that the sex ratio was influenced by the prey type offered to H. miles while, Al Rehiayani and Fouly (2005) reported that the total percentage of daughter females of C. simplex was 61, 54 and 56% feeding on C. rodriguez, M. javanica and T. semipenetran, respectively. Also, Ydergaard et al. (1997) mentioned that thermal factor highly affected female proportion of H. miles which exhibited its highest percentage of daughter females (90.3%) when it was incubated at 25°C in comparison with 15, 20 and 30°C. Each adult female of C. qassimensis was able to lay a total amount of 42.68, 27.70 and 23.83 eggs during 13.58, 11.5 and 8.00 days when it was fed on T. putrescentiae, C. rodriguez and M. incognita, respectively. So, C. qassimensis fed and successfully reproduced when it was provided with these diets. Also, it was previously found that the ascid mite L. scapulatus fed and reproduced on juvenile stages and adults of the nematode Aphelenchus avenae where each female laid an average of 23 eggs during only 6 days (Imbriani and Mankau, 1983). While, Shereef et al. (1980) found that the mean number of eggs per female was 38.6, 16.4 and 47.0 for H. miles fed on bulb mites, collembola and mould mites, respectively. Moreover, the present study showed that the daily deposited eggs per female per day ranged between 2.41 and 3.14 eggs. Similar results were obtained on H. miles by Ydergaard et al. (1997) who mentioned that the mean number of eggs per female per day reached 2.3 eggs when the laelapidae mite fed on a mixture of Bradysia pauper Tuomikoski (Diptera: Sciaridae) and B. tritici Coquillet larvae at 25°C.
Life table parameters of C. qassimensis: The survivorship (Lx curves) of C. qassimensis followed (I) pattern in which survival of immature stages reached 92, 90 and 86% when fed on T. putrescentiae, C. rodriguez and egg masses of M. incognita, respectively. Both parameters Lx and fecundity (Mx) were affected by prey species; however they showed higher values feeding on T. putrescentiae in comparison with C. rodriguez or egg masses of nematode. It was also noticed that Lx values and Mx of L. dentatus and C. simplex were affected by prey species.
Similar results were obtained by Al Rehiayani and Fouly (2005) who mentioned that Lx curves for C. simplex followed I pattern. On the other hand, the mean generation Time (T) and the time required for the population of C. qassimensis to double its number (DT) were affected by prey species. However, T and DT time were shorter when C. qassimensis was fed T. putrescentiae in comparison with C. rodriguez and M. incognita. Similarly, Al-Rehiayani and Fouly (2005) mentioned that T time of C. simplex was shortened when C. simplex fed egg masses of T. semipenetrans and prolonged by feeding on the acarid mite C. rodriguez or egg masses of M. javanica. Net reproductive rate (Ro), the Gross Reproductive Rate (GRR), intrinsic rate of increase (rm) and finite rate of increase (erm) were at their highest values feeding on T. putrescentiae and followed by C. rodriguez while a diet of egg masses of nematode caused the lowest values. Al Rehiayani and Fouly (2005) also found that Ro of C. simplex was lower feeding on M. javanica and T. semipenetran in comparison with C. rodriguez. Also, they noticed that rm and erm of C. simplex were highest on acarid mites in comparison with egg masses of nematodes.
Fouly (1997) reported that Ro values of L. dentatus followed the same trend where they were higher by feeding on acarid mites than feeding on egg masses of root-knot nematode. Moreover, Freira and de Moraes (2007) found that the population of C. paulista fed on T. putrescentiae was estimated to increase about 41.5 times (Ro) in 19.8 days (T) and the rate corresponded to a daily population increase of about 24% (erm) and to the production of 0.217 female per female per day (rm).
Cosmolaelaps qassimensis Fouly and Al-Rehyiani (2014) was collected from debris and rizhosphere of date palm trees growing in Qassim region. The present results showed that C. qassimensis should be provided with acarid mites such as T. putrescentiae or C. rodriguez to obtain the highest rates of offspring. However, the time required for the population of C. qassimensis to double its number were the shortest, it was found that survival percentage, gross reproductive rate, net reproductive rate, intrinsic rate of increase and finite rate of increase were highest when C. qassimensis was subjected to acarid mites as food sources. Generally, C. qassimensis showed good performance in terms of population growth when compared with other Hypoaspidinae at similar conditions.