Research Article
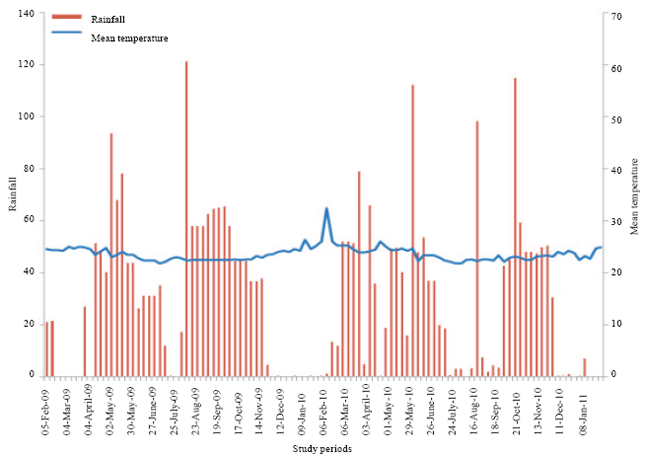
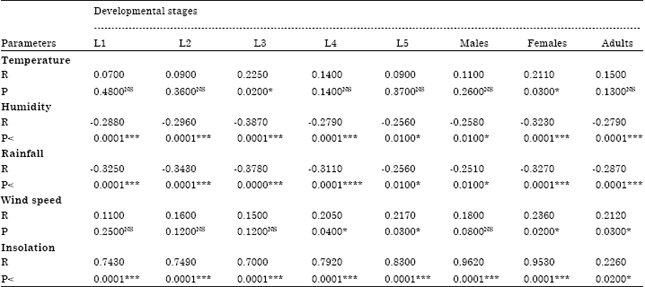
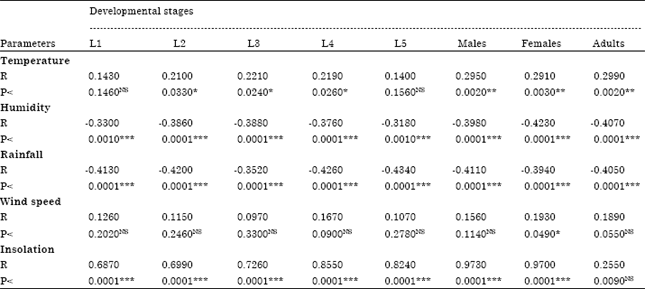
Population Dynamic of Diclidophlebia eastopi Vondracek and Diclidophlebia harrisoni Osisanya, Psyllids Pest of Triplochiton scleroxylon (K. Schum) (Urticales: Sterculiaceae) in Yaounde, Cameroon
Department of Biological Sciences, Laboratory of Zoology, Higher Teachers Training College, University of Yaounde I, P.O. Box 47, Yaounde, Cameroon
Wenceslas Yana
Faculty of Science, University of Bamenda, P.O. Box 39, Bamenda, Cameroon
Joseph Lebel Tamesse
Department of Biological Sciences, Laboratory of Zoology, Higher Teachers Training College, University of Yaounde I, P.O. Box 47, Yaounde, Cameroon