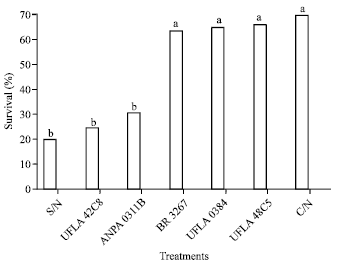
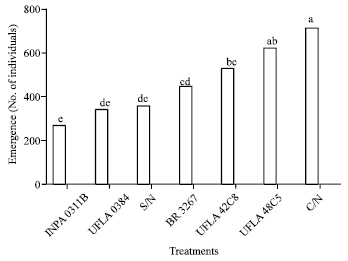
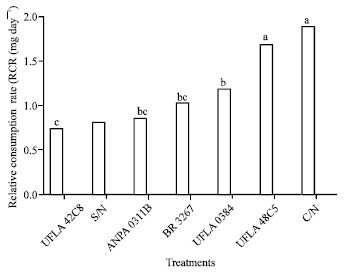
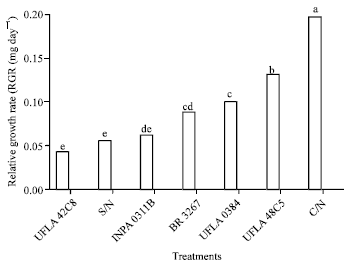
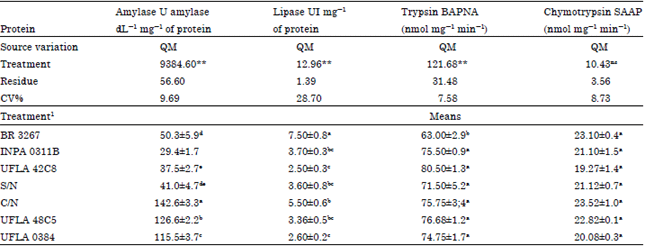
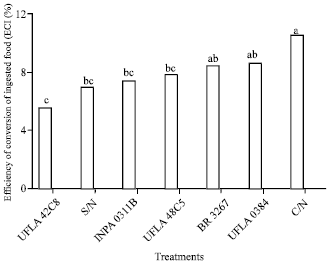
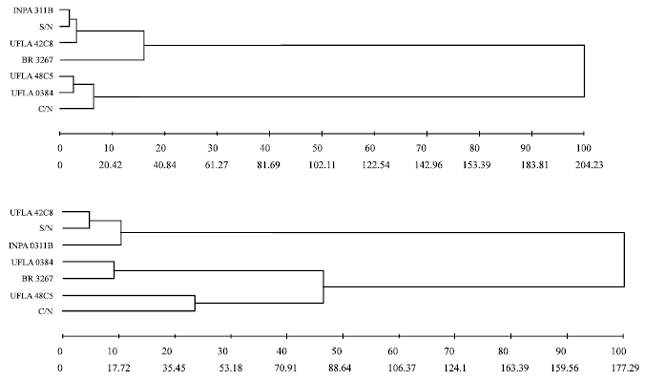
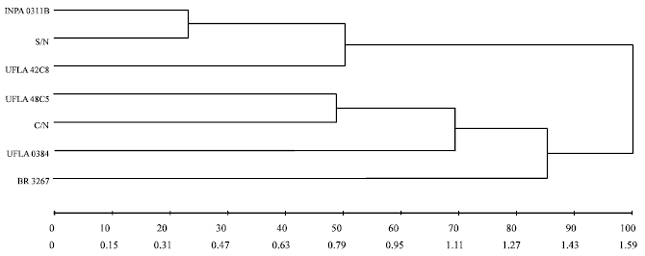
Research Article
Behavior of Callosobruchus maculatus Populations Fed with Vigna unguiculata Grain Cultivated with Diazotrophic Bacteria Strains
Programa de Pos-Graduacao em Agronomia Fitotecnia Universidade Federal do Piaui, Campus Profa. Cinobelina Elvas, Bom Jesus-PI, Brazil
G.S. Carvalho
Departamento de Ciencias da Natureza, Universidade Federal do Piaui, Campus Profa Cinobelina Elvas, Bom Jesus-PI, Brazil
A.C. Mancin
Departamento de Ciencias da Natureza, Universidade Federal do Piaui, Campus Profa Cinobelina Elvas, Bom Jesus-PI, Brazil
L.S. Silva
Departamento de Ciencias da Natureza, Universidade Federal do Piaui, Campus Profa Cinobelina Elvas, Bom Jesus-PI, Brazil
R.S.A. Nobrega
Centro de Ciencias Agrarias, Ambientais e Biologicas, Universidade Federal do Reconcavo da Bahia, Cruz das Almas-BA, Brazi
B.E. Pavan
Programa de Pos-Graduacao em Agronomia Fitotecnia Universidade Federal do Piaui, Campus Profa. Cinobelina Elvas, Bom Jesus-PI, Brazil
K. Maggioni
Programa de Pos-Graduacao em Agronomia Fitotecnia Universidade Federal do Piaui, Campus Profa. Cinobelina Elvas, Bom Jesus-PI, Brazil
E.M. Costa
Departamento de Ciencia do Solo, Universidade Federal de Lavras, Lavras-MG, Brazil