Research Article
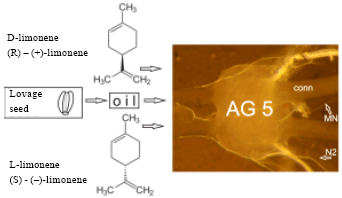
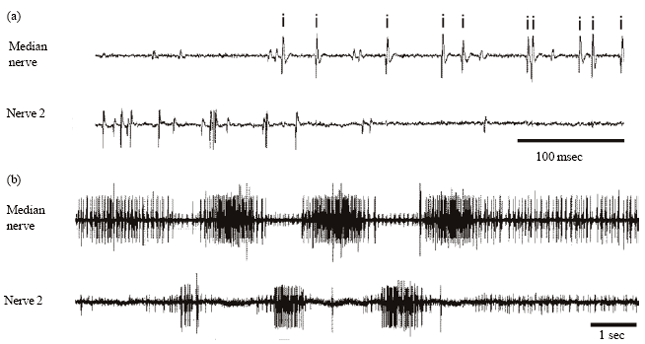
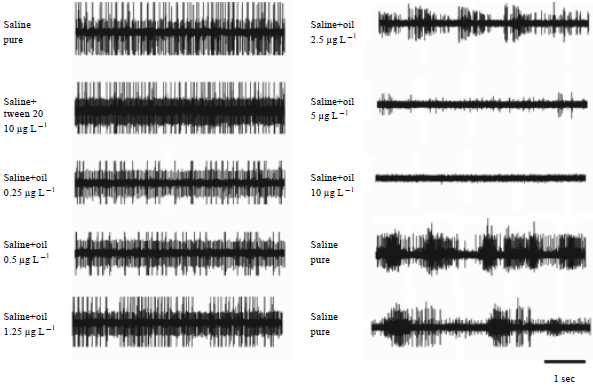
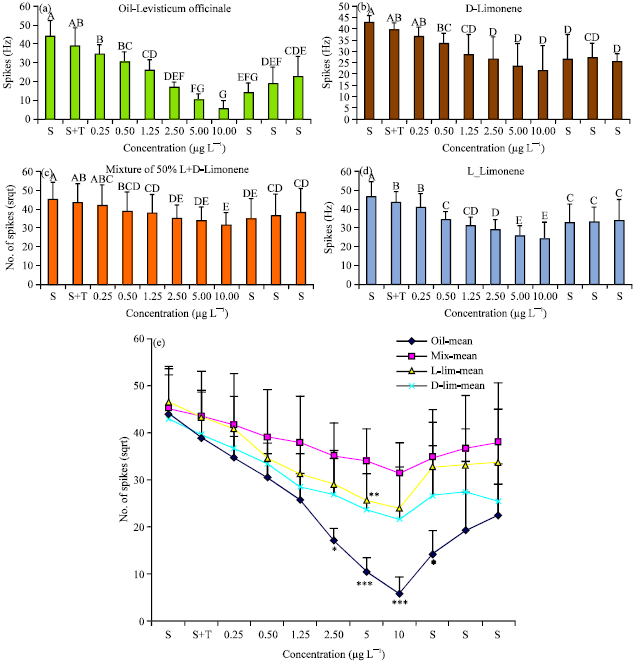
Neural Activity of a Locust Ganglion is Impaired by Limonene and Lovage (Levisticum officinale) Extracts
Department of Plant Protection, Faculty of Agriculture, Behan University, Moshtohor, Egypt
Reinhold Hustert
JFB-Institut of Zoologie, Abt. Neurobiologie, Georg-August-Universitat Gottingen, Germany