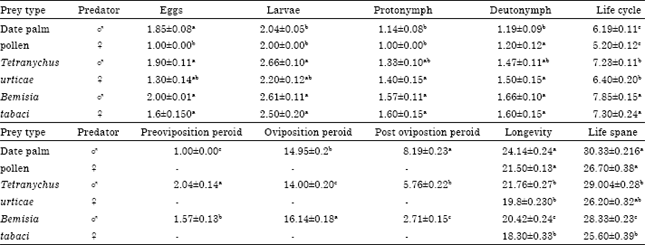
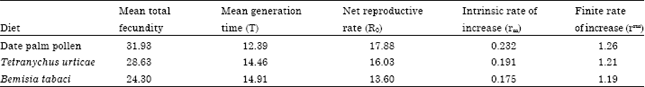
Research Article
Biology and Life Tables of Esieus scutalis (A.-H.) Reared on Different Kinds of Food
Department of Agricultural Zoology, Faculty of Agriculture, Mansoura University, Mansoura 35516, Egypt
O.A. Nassar
Department of Agricultural Zoology, Faculty of Agriculture, Mansoura University, Mansoura 35516, Egypt
M.A. Osman
Department of Agricultural Zoology, Faculty of Agriculture, Mansoura University, Mansoura 35516, Egypt