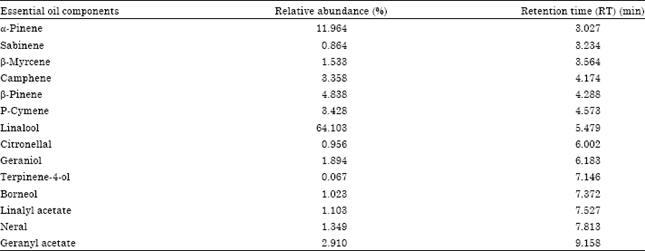
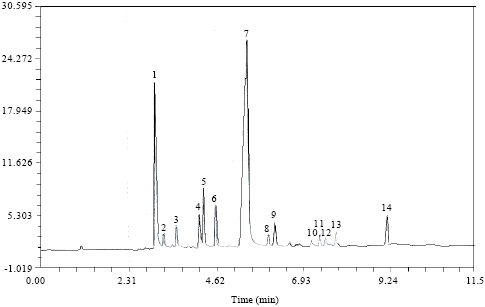
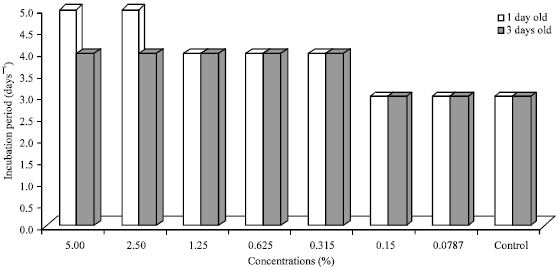
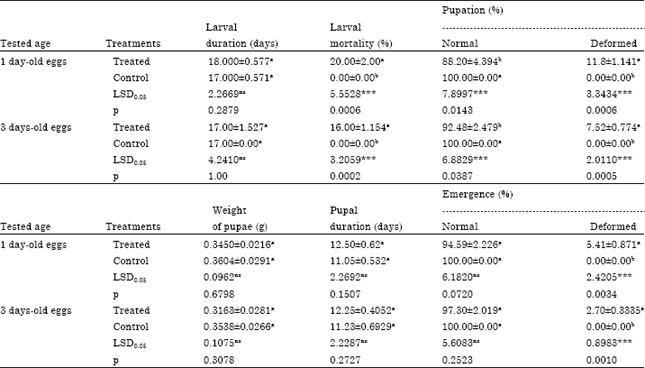
Research Article
Control of Spodoptera littoralis (Boisd.) (Lepidoptera:Noctuidae) and Tetranychus urticae Koch (Acari:Tetranychidae) by Coriander Essential Oil
Plant Protection Research Institute, Giza, Egypt
Hany El-Kawas
Plant Protection Research Institute, Giza, Egypt