Research Article
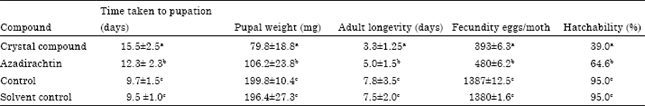
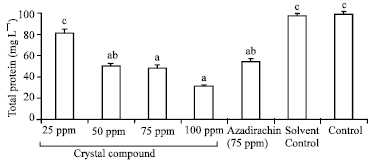
Impact of Novel Crystal Compound 2, 5-diacetoxy-2-benzyl-4,4,6,6-tetramethyl-1,3- Cyclohexanedione on Feeding Physiology and Developmental Indices of Armyworm, Spodoptera litura Fab. (Lepidoptera: Noctuidae)
Department of Zoology, Arignar Anna Government Arts College, Musiri-621 201, Tamil Nadu, India
N. Raja
Department of Biology, Faculty of Natural and Computational Sciences, Post Box 196, University of Gondar, Gondar, Ethiopia
S. Ignacimuthu
Entomology Research Institute, Loyola College, Chennai 600 034, India










savaliya dinesh Reply
how to calculate the relative growth rate.?
pls give me immediately