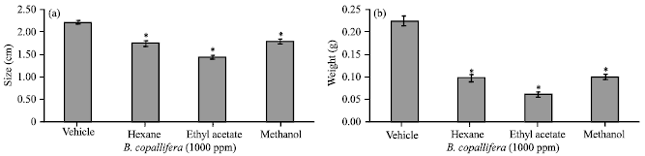
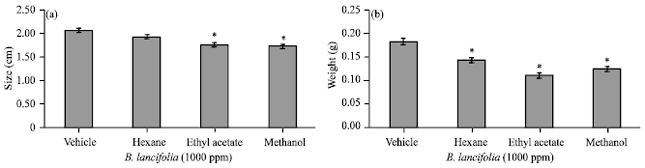
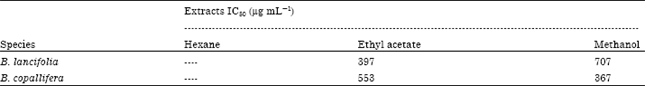
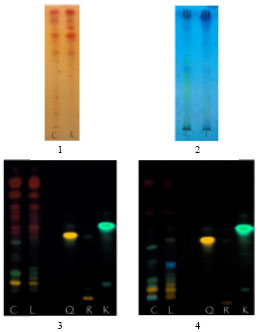
Research Article
Effects of Organic Extracts of Bursera copallifera and B. lancifolia Leaves in the Development of Spodoptera frugiperda
Departamento de Biolog�a Celular, Facultad de Ciencias, Universidad Nacional Aut�noma de Mexico, Ciudad Universitaria, Coyoac�n 04510, Mexico, D.F. Mexico
Jose de Jesus Reguera Serrano
Departamento de Ecolog�a y Recursos Naturales, Facultad de Ciencias, Universidad Nacional Aut�noma de Mexico, Ciudad Universitaria, Coyoac�n 04510, Mexico, D.F. Mexico
Enrique Llanos-Romero
Departamento de Ecolog�a y Recursos Naturales, Facultad de Ciencias, Universidad Nacional Aut�noma de Mexico, Ciudad Universitaria, Coyoac�n 04510, Mexico, D.F. Mexico
Eva Aguirre-Hernandez
Departamento de Ecolog�a y Recursos Naturales, Facultad de Ciencias, Universidad Nacional Aut�noma de Mexico, Ciudad Universitaria, Coyoac�n 04510, Mexico, D.F. Mexico
Josefina Herrera-Santoyo
Departamento de Ecolog�a y Recursos Naturales, Facultad de Ciencias, Universidad Nacional Aut�noma de Mexico, Ciudad Universitaria, Coyoac�n 04510, Mexico, D.F. Mexico
Beatriz Zuniga
Departamento de Ecolog�a y Recursos Naturales, Facultad de Ciencias, Universidad Nacional Aut�noma de Mexico, Ciudad Universitaria, Coyoac�n 04510, Mexico, D.F. Mexico
Beatriz Rodarte
Departamento de Biolog�a Celular, Facultad de Ciencias, Universidad Nacional Aut�noma de Mexico, Ciudad Universitaria, Coyoac�n 04510, Mexico, D.F. Mexico
Luisa Alba-Lois
Departamento de Biolog�a Celular, Facultad de Ciencias, Universidad Nacional Aut�noma de Mexico, Ciudad Universitaria, Coyoac�n 04510, Mexico, D.F. Mexico
Patricia Guevara-Fefer
Departamento de Ecolog�a y Recursos Naturales, Facultad de Ciencias, Universidad Nacional Aut�noma de Mexico, Ciudad Universitaria, Coyoac�n 04510, Mexico, D.F. Mexico