Research Article
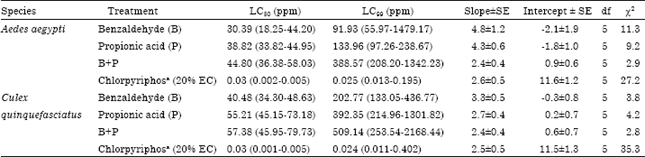
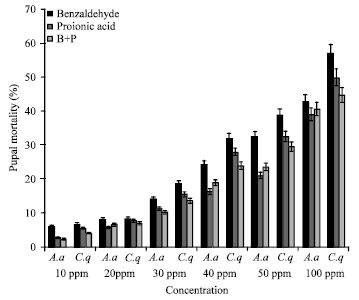
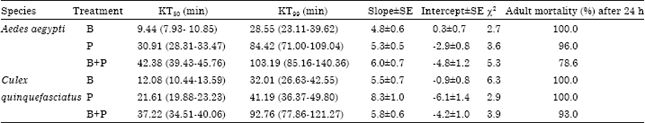
Toxicity of Benzaldehyde and Propionic Acid against Immature and Adult Stages of Aedes aegypti (Linn.) and Culex quinquefasciatus (Say) (Diptera: Culicidae)
Entomology Research Institute Loyola College, Nungambakkam, Chennai-600 034, Tamil Nadu, India
A.D. Reegan
Entomology Research Institute Loyola College, Nungambakkam, Chennai-600 034, Tamil Nadu, India
S. Ignacimuthu
Entomology Research Institute Loyola College, Nungambakkam, Chennai-600 034, Tamil Nadu, India