Research Article
Efficiency of Azadirachtin, A Chitin Synthesis Inhibitor on Growth, Development and Reproductive Potential of Tribolium confusum after Adult Treatment
Department of Biology, Faculty of Science for Girls, King Abdul-aziz University, Jeddah, Saudi Arabia










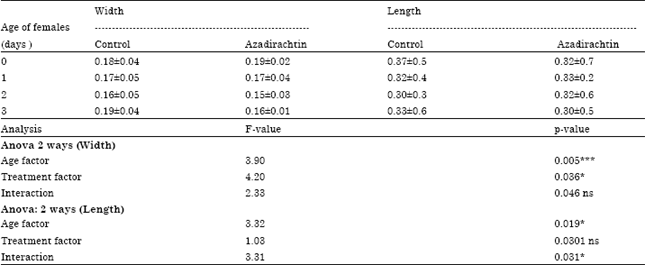
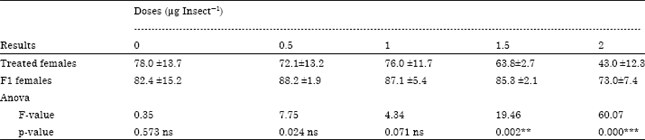
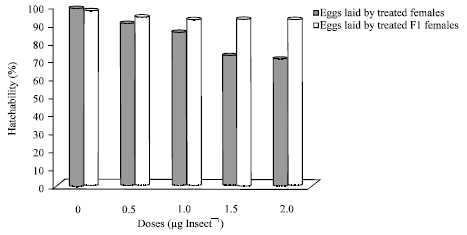

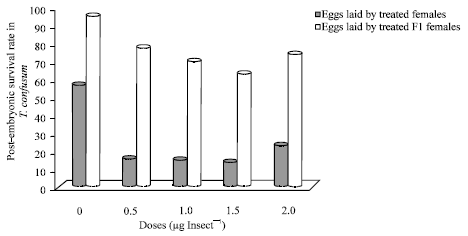
Dr Pandit R.S. Reply
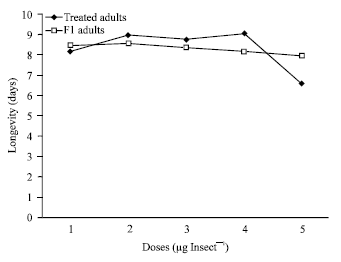
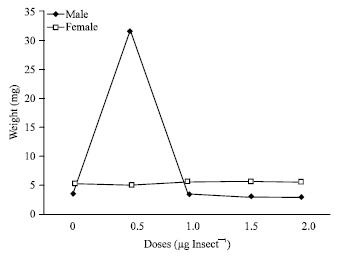
The data published in this paper is very useul and infrmative Efficiency of growth regulators derived from NEEM PLANT IS ONE OF THE BEST remedy against Stored grain pests. for their effective pest management strategy