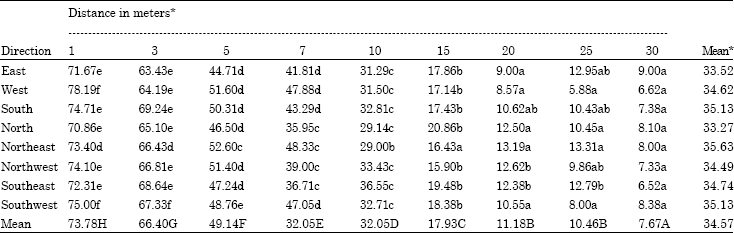
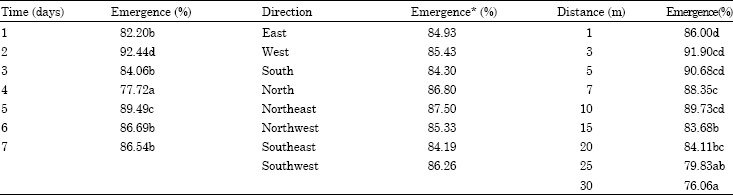
Research Article
Temporal and Spatial Dispersal of Laboratory-Reared Trichogramma chilonis Ishii in Open Field
Division of Crop Protection,Coimbatore-641007, Tamil Nadu, India
R. Balakrishnan
Section of Statistics, Sugarcane Breeding Institute, Coimbatore-641007, Tamil Nadu, India