Research Article
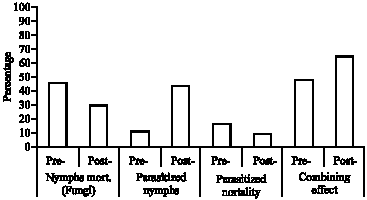
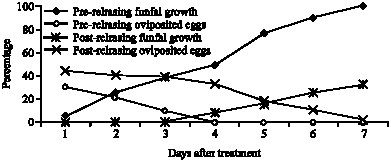
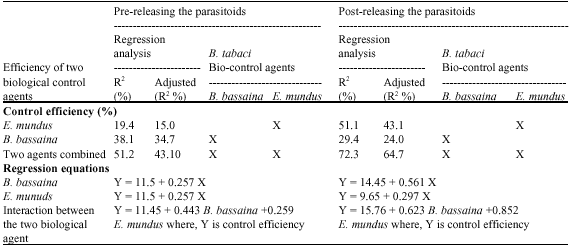
Combining Effect of Beauveria bassaania (Bals.) and Ertemocerus mundus Mercet (Hymenoptera: Aphelinidae) on Sweetpotato Whitefly, Bemisia tabaci Gennadius (Aleyrodidae; Hemiptera)
Department of Crop Production and Protection, College of Agriculture and Veterinary Medicine, Qassim University, P.O. Box 6622, Buraydah 51452, Kingdom of Saudi Arabia