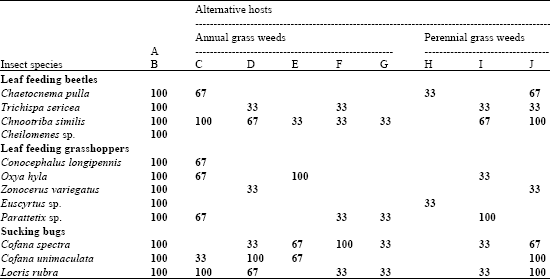
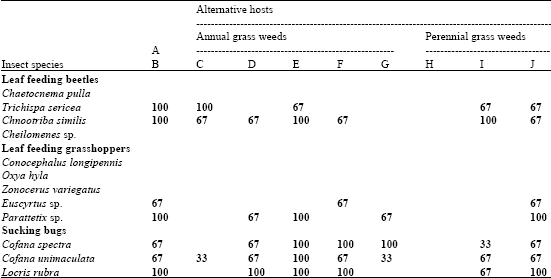
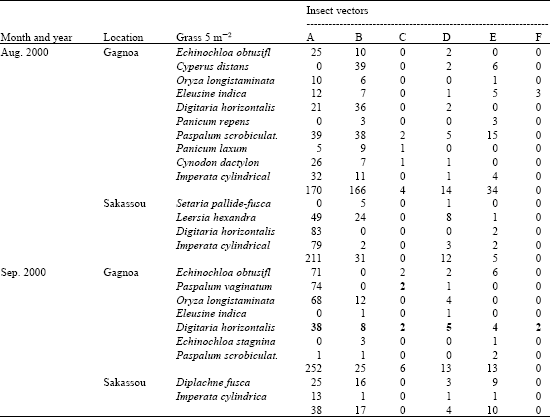
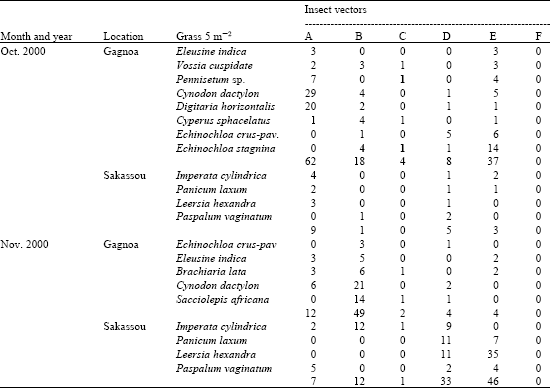
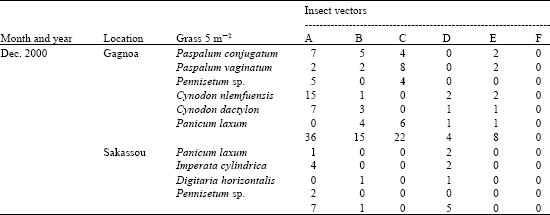
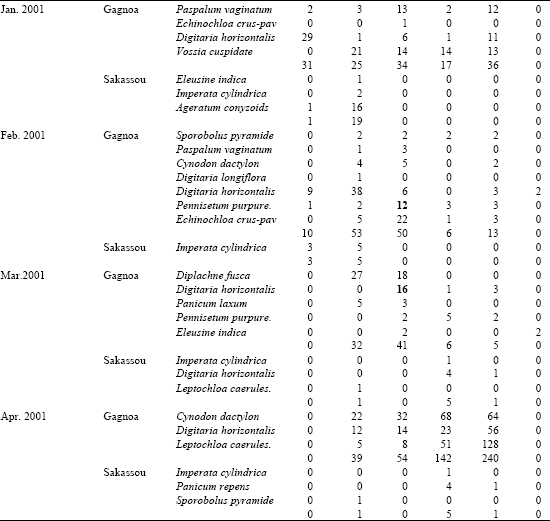
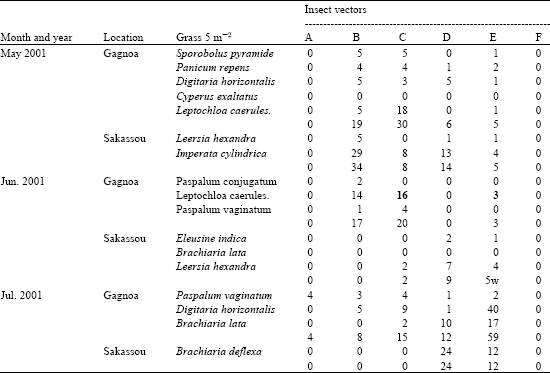
Research Article
New Records of Insect Vectors of Rice Yellow Mottle Virus (RYMV) in Cote d`Ivoire, West Africa
Africa Rice Center (WARDA), P.M.B. 5320, Ibadan Nigeria, West Africa
A.K. Traore
Africa Rice Center (WARDA), 01 B.P. 2551, Bouaké 01, Côte d’Ivoire, West Africa
A.N. Asidi
Africa Rice Center (WARDA), 01 B.P. 2551, Bouaké 01, Côte d’Ivoire, West Africa
Y. Sere
Africa Rice Center (WARDA), 01 B.P. 2031, Cotonou, Benin, West Africa
A. Onasanya
Africa Rice Center (WARDA), 01 B.P. 2031, Cotonou, Benin, West Africa
M.E. Abo
National Cereals Research Institute (NCRI), Badeggi, P.M.B. 8, Bida, Niger State, Nigeria, West Africa